Research Articles
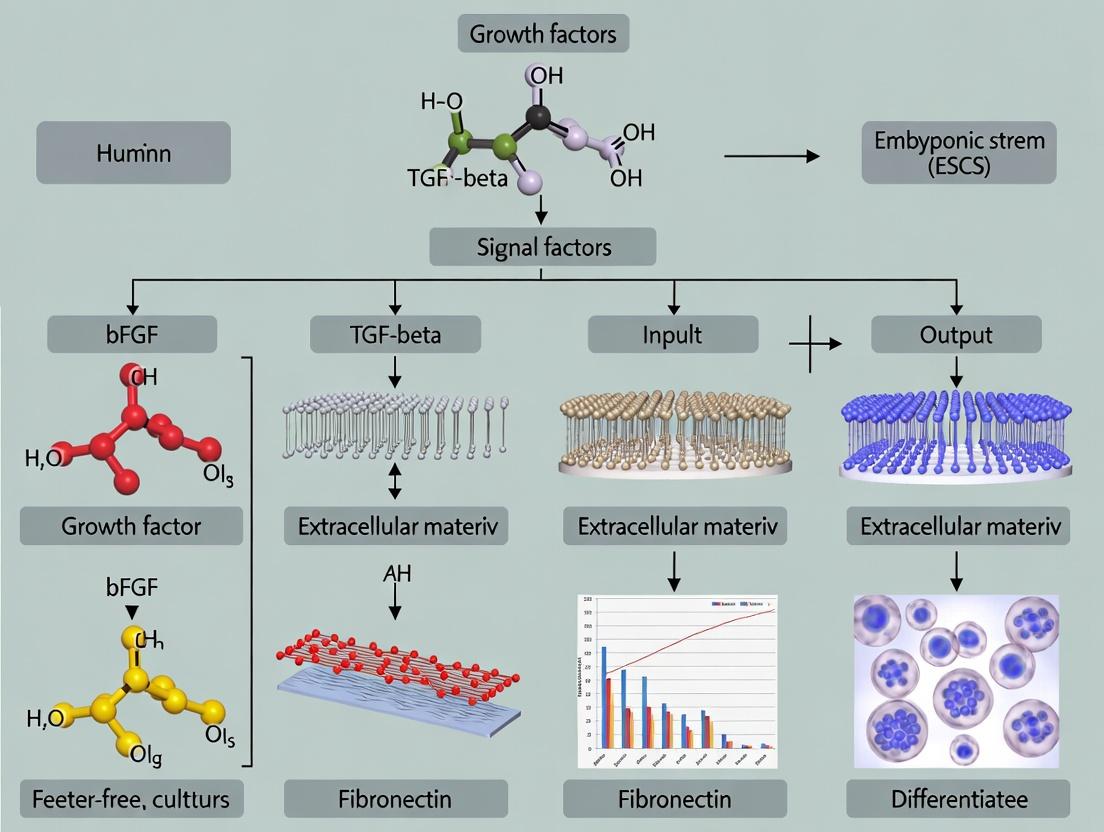
Feeder-Free Human ESC Culture: Protocols, Matrices & Media for Defined Conditions in 2024
This comprehensive guide explores feeder-free culture systems for human embryonic stem cells (hESCs), addressing key needs of researchers and drug developers.
Optimizing Evidence Generation for Novel Therapy Regulatory Submissions: Strategies for RWE, AI, and Adaptive Pathways
This article provides a comprehensive guide for researchers and drug development professionals on navigating the evolving landscape of regulatory evidence generation.
Navigating the Maze: A Researcher's Guide to Solving Data Collection Challenges in Regulatory Frameworks
This article provides a comprehensive roadmap for researchers and drug development professionals grappling with data collection amidst complex and evolving regulatory landscapes.
Beyond the Checklist: A Strategic Framework for Comparative Analysis of Pharmaceutical Validation Parameters in 2025
This article provides researchers, scientists, and drug development professionals with a modern framework for conducting comparative analyses of pharmaceutical validation parameters.
Navigating the Maze: A Practical Guide to Troubleshooting Methodological Issues in Regulatory Comparison Studies
This article provides a comprehensive framework for researchers, scientists, and drug development professionals conducting regulatory comparison studies.
Navigating Multi-Country Medical Device Approvals: A Strategic Guide for Researchers and Developers
This article provides a comprehensive guide for researchers, scientists, and drug development professionals facing the complexities of achieving regulatory approval for medical devices across multiple countries.
Optimizing Regulatory Strategy: A Comparative Framework Analysis for Accelerated Drug Development
This article provides a comprehensive guide for researchers and drug development professionals on leveraging comparative framework analysis to optimize regulatory strategy.
Navigating the Maze: A Strategic Guide to Comparing International Regulatory Frameworks for Drug Development Professionals
This article provides a comprehensive guide for researchers, scientists, and drug development professionals facing the complexities of international regulatory frameworks.
Comparative Analysis in Health Technology Assessment: Methods, Applications, and Global Best Practices
This article provides a comprehensive examination of comparative analysis applications in Health Technology Assessment (HTA) for researchers, scientists, and drug development professionals.
Navigating the New Era of Quality Management: A Strategic Framework for Comparing FDA QMSR and Global Standards
This article provides drug development professionals and researchers with a structured, actionable framework for navigating the pivotal shift in medical device quality management.