Cryopreserved MSCs: A Direct Comparison of TCP vs. Bioreactor Expansion for Clinical Manufacturing
This article provides a critical analysis for researchers and drug development professionals on the impact of expansion methodology—conventional tissue culture plastic (TCP) versus bioreactor systems—on the critical quality attributes of...
Cryopreserved MSCs: A Direct Comparison of TCP vs. Bioreactor Expansion for Clinical Manufacturing
Abstract
This article provides a critical analysis for researchers and drug development professionals on the impact of expansion methodology—conventional tissue culture plastic (TCP) versus bioreactor systems—on the critical quality attributes of mesenchymal stromal cells (MSCs) following cryopreservation. We explore the foundational biological differences induced by each platform, detail scalable manufacturing processes and automated platforms, address key challenges in cryopreservation and optimization, and present a comparative validation of post-thaw cell phenotype, functionality, and therapeutic potency. The synthesis of recent evidence aims to guide the selection and optimization of manufacturing workflows for robust, clinically effective, 'off-the-shelf' MSC-based therapies.
MSCs as Therapeutic Agents: Core Biology and the Imperative for Scalable Expansion
Mesenchymal stem/stromal cells (MSCs) represent a cornerstone of regenerative medicine and cell-based therapy, distinguished by their multipotent differentiation capacity and potent immunomodulatory properties. According to the International Society for Cell & Gene Therapy (ISCT), the minimal defining criteria for human MSCs include: (1) adherence to plastic under standard culture conditions; (2) expression of CD73, CD90, and CD105 surface markers (≥95%), while lacking expression of CD45, CD34, CD14/CD11b, CD79α/CD19, and HLA-DR (≤2%); and (3) ability to differentiate into osteoblasts, adipocytes, and chondrocytes under standard in vitro conditions [1]. These criteria provide a critical foundation for standardizing MSCs across research and clinical applications, ensuring consistent characterization and reliable interpretation of experimental results.
The therapeutic appeal of MSCs extends beyond their trilineage differentiation potential to include robust immunomodulatory effects, trophic capabilities, high in vitro self-renewal ability, and low immunogenicity due to limited expression of HLA-class II and co-stimulatory molecules [2] [3]. These unique characteristics enable both autologous and allogeneic applications, positioning MSCs as promising candidates for treating a diverse range of conditions including graft-versus-host disease, Crohn's disease, myocardial infarction, stroke, and autoimmune disorders [2] [3] [1]. This review examines the fundamental aspects of MSC biology within the specific context of comparing tissue culture polystyrene (TCP) and bioreactor-based expansion systems, with particular emphasis on how cryopreservation impacts cell characteristics and therapeutic potential.
MSCs can be isolated from various tissue sources, each with distinct advantages and limitations for research and clinical applications. The most common sources include bone marrow, adipose tissue, and umbilical cord, though they can also be derived from peripheral blood, dental pulp, placental tissues, and other perinatal tissues [3] [1]. The tissue origin significantly influences MSC characteristics and functionality, necessitating careful selection based on the intended application.
Table 1: Comparison of Primary MSC Tissue Sources
| Tissue Source | Key Markers | Advantages | Limitations | Therapeutic Strengths |
|---|---|---|---|---|
| Bone Marrow (BM-MSCs) | CD73+, CD90+, CD105+ | Gold standard, well-characterized | Invasive collection, low yield | Enhanced osteogenic potential [3] |
| Adipose Tissue (A-MSCs) | CD73+, CD90+, CD105+ | Abundant tissue, minimally invasive harvest | Donor variability | Potent immunomodulation, angiogenic properties [3] [4] |
| Umbilical Cord (UC-MSCs) | CD73+, CD90+, CD105+ | Non-invasive collection, low immunogenicity | Limited cell numbers without expansion | Strong immunomodulation, minimal allogeneic response [3] |
Comparative analyses suggest that A-MSCs may exert more potent immunomodulatory effects than BM-MSCs, while UC-MSCs demonstrate minimal risk of initiating an allogeneic immune response in vivo, making them particularly attractive for allogeneic therapies [3]. Furthermore, source selection influences practical manufacturing considerations, including initial cell numbers, expansion requirements, and scalability – all critical factors when comparing expansion systems.
Immunomodulatory Mechanisms of MSCs
MSCs exert their therapeutic effects through sophisticated immunomodulatory mechanisms that involve both innate and adaptive immune systems. These mechanisms are mediated through two primary pathways: direct cell-to-cell contacts and paracrine activity via secreted factors.
Cell-to-Cell Contact Mechanisms: MSCs directly interact with immune cells through surface molecules to modulate immune responses. They express integrins (α1, α2, α3, α5, α6, αv, β1, β3, β4), intercellular adhesion molecules (ICAM-1, ICAM-2), vascular cell adhesion molecule (VCAM-1), CD72, and CD58 (LFA-3), enabling high-affinity binding to T lymphocytes [1]. Key contact-mediated immunomodulatory interactions include:
- T-cell Regulation: MSCs inhibit naive and memory T-cell responses by upregulating ICAM-1 and VCAM-1, critical for T-cell activation and leukocyte recruitment to inflammation sites [3]. Human placenta-derived MSCs express high levels of PDL1 and PDL2, which inhibit T-cell proliferation by arresting the cell cycle in the G0/G1 phase [3] [1].
- B-cell Regulation: Adipose-derived MSCs increase survival of quiescent B-cells via contact-dependent mechanisms and facilitate B-cell differentiation independently of T-cells [3].
- Monocyte/Macrophage Regulation: MSCs increase TSG-6 production through intercellular contact with pro-inflammatory M1 macrophages, promoting their transformation into anti-inflammatory M2 macrophages [1].
Paracrine Activity Mechanisms: The MSC secretome comprises a diverse repertoire of cytokines, growth factors, chemokines, and extracellular vesicles that collectively modulate immune responses [3]. Key soluble factors include:
- TGF-β/Smad Pathway: TGF-β binds to TGF-βRII, recruiting and phosphorylating TGF-βRI, which triggers phosphorylation of R-Smads (Smad2/3). These form complexes with Smad4 that translocate to the nucleus to regulate transcription of immune suppression genes like FOXP3, the master regulator of Tregs [1].
- IDO and PGE2 Pathways: Indoleamine-pyrrole 2,3-dioxygenase (IDO) and prostaglandin E2 (PGE2) suppress dendritic cell-mediated Th2 priming and inhibit T helper 17 cell (Th17) differentiation while promoting Treg induction [3] [1].
- Extracellular Vesicles: MSC-derived exosomes and microvesicles carry immunoregulatory molecules including miRNAs (e.g., miR-146a-5p) that directly inhibit eosinophil activation and IgE production in allergic inflammation models [3] [1].
The following diagram illustrates the coordinated signaling pathways through which MSCs mediate their immunomodulatory effects:
Experimental Comparison: TCP vs. Bioreactor Expansion Systems
The expansion methodology significantly influences MSC characteristics, requiring careful consideration of manufacturing platforms. The two primary systems for MSC expansion are tissue culture polystyrene (TCP) flasks and hollow fiber bioreactors (HFB), each with distinct operational parameters and scalability profiles.
Table 2: Manufacturing System Comparison: TCP vs. Hollow Fiber Bioreactor
| Parameter | TCP Flask System | Hollow Fiber Bioreactor (HFB) |
|---|---|---|
| Scalability | Limited, requires increasing flask numbers | High, integrated scalable platform |
| Labor Intensity | High, numerous manual operations | Automated, reduced hands-on time |
| Process Openness | Multiple open procedures (e.g., 54,400 for clinical trial) [2] | Functionally closed system (e.g., 133 procedures) [2] |
| Contamination Risk | Higher due to open procedures | Substantially reduced [2] |
| Cell Harvesting | Enzymatic digestion at each passage | Automated harvest capability |
| Initial Investment | Lower cost, standard equipment | High capital investment |
| Process Monitoring | Manual sampling | Integrated monitoring capabilities |
The Quantum Cell Expansion System (Terumo BCT), a widely implemented hollow fiber bioreactor, represents a Good Manufacturing Practice (GMP)-compliant platform for adherent cell expansion [2] [5]. This system utilizes a hollow fiber bioreactor within a self-contained unit with computerized control of medium perfusion rates, harvest parameters, and other critical processes [2]. Compared to TCP-based methods, the Quantum system demonstrated the ability to generate target cell numbers (2.0×10⁸ cells) in 9-fewer days with half the number of passages [2].
Experimental Protocols for Comparative Studies
TCP Flask Expansion Protocol: Bone marrow mononuclear cells (BMMCs) are isolated from bone marrow aspirate using Ficoll density gradient separation on a Sepax cell separation device [2]. BMMCs are resuspended in specialized media containing 5% platelet lysate, 2.1 units/mL heparin, 2mM GlutaMax, and 10mM N-acetylcysteine, then plated at 5×10⁵ cells/cm² in T-175 cm² flasks (designated Passage 1) [2]. Upon reaching 70-80% confluence, the monolayer is washed and harvested using TrypLE Select, with cells split 1:4 into new flasks at seeding densities of 2×10³-2×10⁴ cells/cm² [2]. Cells are maintained at 37°C with 5% CO₂, typically requiring 3-5 passages before harvest and cryopreservation in media containing 5% human serum albumin, 10% DMSO, and 85% Plasmalyte [2].
Hollow Fiber Bioreactor Expansion Protocol: The bioreactor system is primed with phosphate-buffered saline before coating with 5mg of fibronectin for 4-18 hours [2]. After coating, the system is washed and filled with culture media. Unprocessed bone marrow (25mL) is transferred to a bioreactor cell inlet bag, filtered through a 200μm filter, and loaded into the bioreactor [2]. Cells are allowed to attach for 48 hours before initiating continuous media perfusion, starting at 0.1 mL/minute and progressively increasing based on lactate concentration (doubling rate when lactate reaches 4mM) [2]. Cells are typically harvested after approximately 21 days of culture [2].
Cryopreservation Protocol for Both Systems: For both expansion methods, cells are cryopreserved using controlled-rate freezing in cryopreservation medium containing a final concentration of 5% human serum albumin (HSA), 10% dimethyl sulfoxide (DMSO), and 85% Plasmalyte [2]. Post-thaw analyses are typically conducted after 24 hours of recovery culture [4].
The following diagram illustrates the comparative experimental workflow for both expansion systems:
Comparative Performance Data Post-Cryopreservation
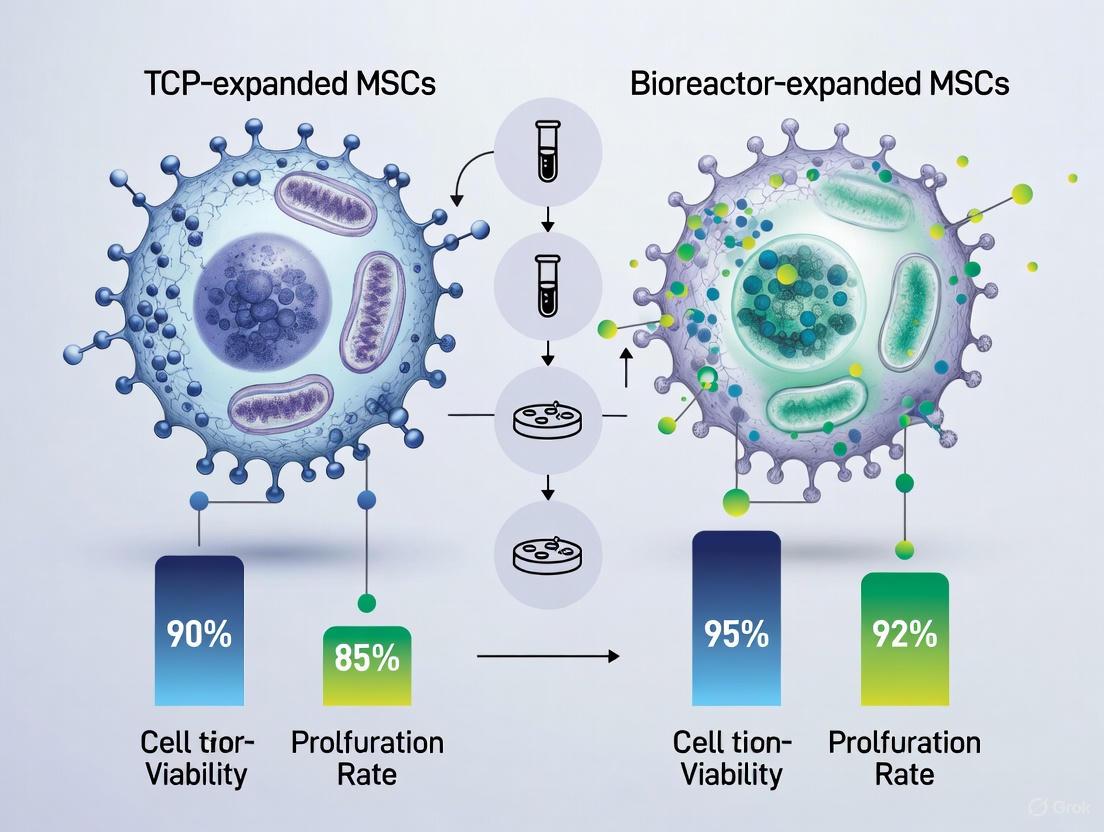
Post-thaw analyses reveal critical differences in MSC characteristics depending on the expansion platform. A 2024 study directly compared cryopreserved adipose-derived stem cells (ASCs) expanded in TCP versus HFB systems, ensuring comparable population doublings between platforms [4].
Table 3: Post-Thaw Characterization of TCP vs. HFB-Expanded MSCs
| Parameter | TCP-Expanded MSCs | HFB-Expanded MSCs | Significance |
|---|---|---|---|
| CD73 Expression | >95% | >95% | No significant difference [4] |
| CD90 Expression | >95% | >95% | No significant difference [4] |
| CD105 Expression | Significant decrease to ~75% post-thaw [4] | Maintained >95% post-thaw [4] | p<0.05 |
| CD274 (PD-L1) Expression | Increased ~48% post-thaw [4] | Pre-freeze: significantly lower; Post-thaw: comparable to TCP [4] | Significant change with cryopreservation |
| Viability Post-Thaw | >90% | >90% | TCP demonstrated greater robustness [4] |
| Trilineage Differentiation | Maintained (adipogenic, osteogenic, chondrogenic) [4] | Maintained (adipogenic, osteogenic, chondrogenic) [4] | No significant difference |
| Population Doublings Time | Longer (reference: +9 days to reach 2.0×10⁸ cells) [2] | Shorter (reference: 21 days to reach 6.6×10⁸ cells) [2] | p<0.05 |
| Subpopulation Heterogeneity | Less variable after thawing [4] | More variable after thawing [4] | Distinct expansion-specific profiles |
The differential CD105 expression pattern is particularly noteworthy, as this marker is part of the ISCT minimal criteria for MSC definition [1]. The significant decrease in TCP-expanded cells post-thaw suggests potential phenotypic instability during the freeze-thaw process in traditionally expanded cells. Both systems maintained trilineage differentiation capacity post-thaw, confirming retention of fundamental MSC characteristics despite cryopreservation [4].
Beyond surface markers, functional assessments demonstrate that MSCs from both expansion systems retain therapeutic efficacy. In vitro wound healing assays showed no statistical differences in the effects on fibroblast migration between TCP and HFB-expanded ASCs after cryopreservation [4]. More importantly, Quantum-expanded MSCs demonstrated therapeutic activity in an ischemic stroke rat model, confirming functional competence after bioreactor expansion [2].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful MSC research requires carefully selected reagents and specialized equipment. The following table details essential materials for comparative expansion and cryopreservation studies:
Table 4: Essential Research Reagents and Equipment for MSC Studies
| Category | Specific Reagent/Equipment | Function/Application | Examples from Studies |
|---|---|---|---|
| Culture Media Components | Platelet Lysate | Serum replacement for GMP-compliant culture | 5% in D-5 medium [2] |
| Heparin | Prevent coagulation in platelet lysate media | 2.1 units/mL [2] | |
| GlutaMax | Stable source of L-glutamine | 2mM in culture medium [2] | |
| N-acetylcysteine | Antioxidant protection | 10mM in culture medium [2] | |
| Dissociation Reagents | TrypLE Select | Enzymatic cell dissociation | Harvesting from TCP flasks [2] |
| Matrix Proteins | Fibronectin | Enhance cell adhesion to bioreactor fibers | 5mg for bioreactor coating [2] |
| Cryopreservation Components | Dimethyl Sulfoxide (DMSO) | Cryoprotective agent | 10% in cryopreservation medium [2] |
| Human Serum Albumin (HSA) | Protein stabilizer during freezing | 5% in cryopreservation medium [2] | |
| Plasmalyte | Base solution for cryopreservation medium | 85% in cryopreservation medium [2] | |
| Expansion Equipment | Tissue Culture Polystyrene Flasks | Standard 2D expansion platform | T-175 cm² flasks [2] |
| Hollow Fiber Bioreactor | 3D expansion in automated system | Quantum Cell Expansion System [2] [5] | |
| Monitoring Equipment | Glucose/Lactate Meters | Metabolic monitoring during expansion | Aviva Accu-Chek, LactatePlus [2] |
| Characterization Tools | Flow Cytometry | Immunophenotype analysis | CD73, CD90, CD105 quantification [4] |
Advanced research platforms continue to emerge, including microfluidic bioreactors that allow precise control of shear stress during expansion and cryopreservation. Studies demonstrate that regulated shear flow (0.002-0.004 μbar) enhances focal point adhesions and improves post-thaw recovery of adherent MSCs [6]. Additionally, dynamic 3D culture systems using spinner flasks or rotating wall vessels promote spheroid formation with enhanced therapeutic potential compared to static culture [7].
The comparative analysis of TCP and bioreactor expansion systems for MSCs reveals a complex landscape with distinct advantages for each platform. TCP flask systems offer accessibility and lower initial investment but present significant limitations in scalability, labor requirements, and process control. Conversely, hollow fiber bioreactor systems like the Quantum provide automated, closed-system expansion with enhanced scalability and reduced contamination risk, albeit with higher capital investment and technical complexity.
Critical evaluation of post-thaw characteristics demonstrates that both expansion methods can yield clinically relevant MSCs that retain core functionalities, including trilineage differentiation capacity and immunomodulatory potential. However, important differences emerge in phenotypic stability, particularly regarding CD105 expression patterns after cryopreservation. These findings underscore the necessity of selecting expansion methodologies aligned with specific research or clinical objectives, considering both practical manufacturing constraints and critical quality attributes of the final cellular product.
As the field advances, integration of novel technologies such as microfluidic systems, dynamic 3D culture platforms, and advanced cryopreservation protocols with controlled shear stress will further enhance our ability to manufacture functionally optimized MSCs. Standardization of expansion and cryopreservation protocols across platforms remains essential for meaningful comparison of research outcomes and clinical efficacy. The continued refinement of MSC manufacturing platforms will undoubtedly accelerate the translation of these promising cellular therapies from bench to bedside.
The global Mesenchymal Stem Cells (MSCs) market is experiencing substantial growth, valued at USD 3.82 billion in 2024 and projected to reach USD 9.08 billion by 2035, with a compound annual growth rate (CAGR) of 8.20% [8]. This expansion is fueled by the increasing therapeutic application of MSCs in treating orthopedic conditions, autoimmune diseases, cardiovascular disorders, and graft-versus-host disease (GvHD). To meet this rising clinical demand, the field is transitioning from traditional, small-scale autologous therapies toward allogeneic "off-the-shelf" products that enable large-scale production [8] [9]. This paradigm shift necessitates the development of robust, scalable expansion bioprocesses that consistently yield high-quality, clinically relevant MSCs, particularly after the critical step of cryopreservation.
The choice of expansion system is a critical decision in biomanufacturing, balancing practical considerations with the imperative to maintain therapeutic cell potency. Traditional tissue culture polystyrene (TCP) flasks are favored for their cost-effectiveness and versatility but are labor-intensive and hinder scalability due to their manual handling requirements [4]. In contrast, hollow fiber bioreactor (HFB) systems offer automation, superior scalability, and enhanced consistency, making them ideal for large-scale clinical manufacturing, albeit with higher initial costs and technical complexity [4]. As most clinical protocols utilize cryopreserved cells, understanding how these expansion systems influence MSC characteristics post-thaw is essential for guiding biomanufacturing decisions. This review objectively compares TCP and bioreactor-expanded MSCs after cryopreservation, providing researchers and drug development professionals with critical experimental data to inform protocol development.
Comparative Analysis of Expansion and Cryopreservation Systems
Key Characteristics of Expansion Systems
Table 1: Comparison of MSC Expansion Systems
| Feature | Tissue Culture Polystyrene (TCP) | Hollow Fiber Bioreactor (HFB) |
|---|---|---|
| Scalability | Limited, surface area-dependent | High, designed for large-scale production |
| Labor Intensity | High, requires manual handling and passaging | Low, automated and closed system |
| Process Consistency | Prone to variability due to manual operations | High reproducibility and control |
| Initial Cost | Low | High |
| Cell Harvesting | Requires enzymatic digestion (e.g., trypsin) | Can be less disruptive, but system-dependent |
| Physiological Mimicry | Low, 2D, non-physiological shear stress | Better, can provide more 3D-like conditions |
Experimental Workflow for Comparative Studies
A standardized experimental approach is crucial for a direct comparison between expansion systems. One robust methodology involves expanding MSCs from the same donor in both TCP and HFB systems, ensuring comparable population doublings despite different passaging schedules [4].
Diagram: Experimental Workflow for Comparing TCP vs. HFB
Impact on Immunophenotype and Subpopulations Post-Cryopreservation
A critical quality attribute for MSCs is their immunophenotype, defined by the International Society for Cellular Therapy (ISCT) as ≥95% expression of CD73, CD90, and CD105, and ≤2% expression of hematopoietic markers (CD34, CD45, CD14, CD19, HLA-DR) [10] [11]. Studies indicate that while cryopreservation generally preserves most markers, the expansion system can influence specific changes.
Table 2: Immunophenotypic Changes After Cryopreservation by Expansion System
| Surface Marker | TCP Pre-Freeze | TCP Post-Thaw | HFB Pre-Freeze | HFB Post-Thaw | Key Change |
|---|---|---|---|---|---|
| CD73 / CD90 | >95% [4] | >95% [4] | >95% [4] | >95% [4] | Preserved in both systems |
| CD105 | >95% [4] | ~75% [4] | >95% [4] | >95% [4] | Significant drop in TCP |
| CD274 (PD-L1) | Higher [4] | Increased ~48% [4] | Significantly lower [4] | Comparable to TCP [4] | Difference balanced post-thaw |
| CD34 | <9% [4] | Low [4] | <9% [4] | Low [4] | Difference increased post-thaw |
| CD49d (α4-integrin) | Not Reported | Decreased [12] | Not Reported | Decreased [12] | Decreased post-thaw, may affect cell retention |
The freeze-thaw process can also drive differential changes in immunophenotypical subpopulations. Research shows that the proportion of triple-positive (CD73+, CD90+, CD105+) cells, the most abundant population, decreases significantly in TCP-expanded cells after thawing, while HFB-expanded cells show greater stability [4]. Furthermore, TCP and HFB systems inherently support different subpopulations, influencing the heterogeneity of the final product [4].
Functional Characteristics: Potency, Viability, and Differentiation
For clinical application, the retention of functional potency after thawing is paramount. Key functional assays include trilineage differentiation, colony-forming unit (CFU) potential, proliferation capacity, and viability.
Table 3: Functional Characteristics of Cryopreserved MSCs by Expansion System
| Functional Assay | TCP Expanded | HFB Expanded | Significance |
|---|---|---|---|
| Trilineage Differentiation | Preserved post-thaw [4] | Preserved post-thaw [4] | No statistical difference between systems |
| Colony-Forming Unit (CFU) | Baseline [4] | Appeared higher, not statistically significant [4] | Trend warrants further investigation |
| Proliferation / Growth Kinetics | No significant difference [4] | No significant difference [4] | Comparable performance post-thaw |
| Cell Viability Post-Thaw | >90%, more robust [4] | >90% [4] | High viability in both, TCP potentially more robust |
| Fibroblast Migration (Wound Healing) | Supported [4] | Supported [4] | No statistical difference in paracrine effect |
The overall conclusion from functional studies is that the freeze-thaw process does not fundamentally compromise the core functional capabilities of MSCs expanded in either TCP or HFB systems [4]. This functional preservation is also observed in other MSC preparations, such as bone marrow aspirate concentrate (BMAC), where short-term cryopreservation did not negatively affect MSC proliferation, multilineage differentiation, or efficacy in an osteoarthritis rat model [13].
Advanced Culture Systems and Future Outlook
While TCP and HFB represent current standards, research into advanced 3D culture platforms is ongoing. Hydrogel-based systems, such as the Bio-Block platform, are designed to better mimic the native tissue environment. In comparative studies, ASCs cultured in Bio-Blocks showed superior outcomes versus 2D TCP, spheroids, or Matrigel, including:
- ~2-fold higher proliferation
- 30-37% reduced senescence
- 2-3-fold decrease in apoptosis
- Enhanced retention of stem-like markers (LIF, OCT4, IGF1) and trilineage differentiation capacity
- Preservation of secretome protein and increased production of potent extracellular vesicles (EVs) [14]
These findings highlight the critical influence of the culture system on MSC phenotype and function and point toward a future of more sophisticated, biomimetic biomanufacturing platforms.
The future market landscape for MSCs is dynamic, characterized by several key trends:
- A shift from autologous to allogeneic products: Allogeneic MSCs accounted for 54.4% of the market in one segmentation, driven by their "off-the-shelf" potential and industrial scalability [8] [9].
- Convergence with enabling technologies: Integration of MSCs with biomaterials, gene editing (e.g., CRISPR), and artificial intelligence for potency prediction is creating new therapeutic avenues [8] [9].
- Rise of cell-free therapies: MSC-derived exosomes are gaining significant traction as a stable, low-immunogenicity alternative to whole-cell therapies [8] [9].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for MSC Expansion and Characterization
| Reagent / Material | Function in Research | Application Example |
|---|---|---|
| RoosterNourish MSC-XF Medium | Chemically-defined, xeno-free medium for MSC expansion | Used for culture and expansion of Adipose-derived MSCs (ASCs) in 3D culture studies [14] |
| Collagenase Type I | Enzyme for tissue dissociation and isolation of primary MSCs | Digestion of adipose tissue to isolate Stromal Vascular Fraction (SVF) and ASCs [12] |
| Ficoll Gradient | Density gradient medium for isolation of mononuclear cells | Separation of mononuclear cells from bone marrow aspirate or other sources [13] |
| Dimethyl Sulfoxide (DMSO) | Cryoprotectant agent (CPA) for cell freezing | Standard component (e.g., 10% concentration) in cryopreservation solutions for MSCs [4] [12] |
| Flow Cytometry Antibodies (CD73, CD90, CD105, CD34, CD45, HLA-DR) | Immunophenotypic characterization of MSCs | Verification of MSC identity per ISCT criteria and analysis of subpopulations [4] [10] [11] |
| Tri-lineage Induction Kits (Osteo, Chondro, Adipo) | Functional potency assays for MSC differentiation | In vitro demonstration of multipotency; staining with Alizarin Red, Alcian Blue, Oil Red O [4] [12] |
| Poly(Vinylidene Fluoride) (PVDF) Substrates | Electroactive cell culture substrate for directed differentiation | Studying the effect of electroactivity and surface charge on MSC differentiation, particularly osteogenesis [15] |
The comparative analysis of TCP and bioreactor-expanded MSCs reveals a nuanced landscape for clinical biomanufacturing. TCP systems offer a cost-effective and versatile platform for research and small-scale applications, but their manual nature introduces variability, and they can be susceptible to specific post-thaw phenotypic changes, such as a loss of CD105 expression. HFB systems address the critical needs of scalability and reproducibility for commercial therapies, demonstrating robust post-thaw performance with stable immunophenotype.
The experimental data confirms that cryopreservation is a viable strategy for creating "off-the-shelf" MSC products, as it does not fundamentally abolish the core functional properties of the cells, regardless of the expansion system. The choice between TCP and HFB ultimately depends on the stage of development and clinical objectives: TCP suffices for proof-of-concept and early-phase trials, while HFB and other advanced bioreactor systems are indispensable for the large-scale, consistent production required for late-phase trials and commercial distribution. As the field advances, the integration of novel, biomimetic 3D culture platforms and a growing focus on allogeneic, cryopreserved products will continue to shape the clinical demand and production paradigms for MSC-based therapies.
Mesenchymal stem cells (MSCs) have emerged as powerful tools in regenerative medicine and cell-based therapies, with over 1,670 clinical trials currently exploring their potential [16]. These multipotent cells, capable of differentiating into osteoblasts, chondrocytes, and adipocytes, exert their therapeutic effects primarily through paracrine signaling and immunomodulation [17]. The clinical translation of MSC therapies faces a critical manufacturing challenge: producing the vast quantities of cells required for therapeutic doses (ranging from 10⁶ to 10⁹ cells per patient) while maintaining consistent quality and functionality [18]. This challenge is further complicated by the need for cryopreservation, which enables "off-the-shelf" availability of cell therapies but may impact cell characteristics [4] [19].
Two primary expansion systems have emerged to address these manufacturing needs: traditional tissue culture polystyrene (TCP) flasks and bioreactor systems. TCP-based cultures represent the established workhorse of laboratory-scale cell expansion, valued for their simplicity and cost-effectiveness. In contrast, bioreactor systems, including hollow fiber bioreactors (HFB) and stirred-tank reactors with microcarriers, offer automated, scalable solutions for clinical-grade manufacturing [4] [18]. The choice between these platforms has profound implications for scalability, reproducibility, and ultimately, the clinical efficacy of the final cell product, particularly after the critical freeze-thaw process [4] [20]. This comparison guide objectively evaluates the performance of TCP versus bioreactor-expanded MSCs following cryopreservation, providing researchers with experimental data to inform their manufacturing decisions.
Platform Comparison: Fundamental Characteristics and Workflows
The fundamental differences between TCP and bioreactor expansion systems extend beyond simple scale considerations to encompass distinct operational parameters, environmental controls, and processing workflows that significantly impact the final cell product.
TCP Systems represent the conventional approach to MSC expansion, utilizing two-dimensional plastic surfaces for cell adhesion and proliferation. These systems are characterized by their operational simplicity and low initial investment, making them accessible for research and small-scale applications. However, TCP systems are labor-intensive, requiring extensive manual handling for feeding, passaging, and harvesting, which introduces variability and limits scalability [4]. The surface area-to-volume ratio is relatively low, necessitating numerous flasks to achieve high cell yields. Environmentally, TCP systems offer limited control over critical parameters such as pH, dissolved oxygen, and metabolite concentrations, which can fluctuate significantly between medium exchanges [20]. The workflow typically involves serial passaging until sufficient cell numbers are obtained, followed by harvesting and cryopreservation.
Bioreactor Systems, particularly hollow fiber bioreactors (HFB) and stirred-tank reactors with microcarriers, provide a three-dimensional environment for MSC expansion with enhanced process control. These systems excel in scalability and consistency, making them ideal for clinical-grade manufacturing where reproducibility is paramount [4] [18]. HFBs consist of thousands of hollow capillary fibers that provide a substantial surface area for cell growth within a compact footprint, while simultaneously allowing for continuous medium perfusion and precise regulation of the cellular microenvironment [4]. Stirred-tank bioreactors with microcarriers suspend small beads in culture medium, offering efficient scalability through increased surface area per volume [18]. The primary limitations of bioreactor systems include high initial costs, technical complexity, and the need for specialized expertise [4]. The workflow typically involves a single expansion phase within the bioreactor, followed by harvesting and cryopreservation, with some systems incorporating automated cell retention and harvesting mechanisms [18].
Table 1: Fundamental Characteristics of TCP and Bioreactor Expansion Systems
| Characteristic | TCP Systems | Bioreactor Systems |
|---|---|---|
| Scalability | Limited, requires multiple flasks | High, suitable for large-scale production |
| Process Control | Limited control over pH, O₂, metabolites | Precise control over critical parameters |
| Labor Requirements | High, manual handling | Low, automated systems |
| Initial Investment | Low | High |
| Reproducibility | Variable due to manual operations | High consistency and standardization |
| Surface Area Efficiency | Low surface area-to-volume ratio | High surface area-to-volume ratio |
| Environmental Mimicry | 2D, non-physiological | 3D, more physiologically relevant |
Diagram Title: Experimental Workflow for TCP vs. Bioreactor MSC Expansion
Comparative Experimental Data: Functional and Phenotypic Outcomes Post-Cryopreservation
Recent comparative studies have provided quantitative insights into how expansion systems influence MSC characteristics after cryopreservation. A 2024 study directly compared cryopreserved adipose-derived stem cells (ASCs) expanded in TCP versus hollow fiber bioreactor (HFB) systems, ensuring comparable population doublings between platforms [4]. The findings reveal both conserved functionalities and notable phenotypic differences following the freeze-thaw process.
Immunophenotypic and Functional Characteristics
The immunophenotypic analysis demonstrated that most surface markers remained consistent between systems after thawing. Both TCP and HFB-expanded cells maintained high expression (>95%) of CD73 and CD90, key MSC markers defined by ISCT criteria [4] [17]. However, a statistically significant decrease in CD105 expression was observed specifically in TCP-expanded cells after thawing, dropping from >95% to only 75% positive cells [4]. CD274 (PD-L1) showed a distinctive pattern: while significantly less expressed on HFB-expanded cells before freezing, the proportion of CD274 positive cells became comparable to TCP cells after thawing, increasing by nearly 48% [4].
Despite these phenotypic variations, functional characteristics remained remarkably consistent between systems. Both TCP and HFB-expanded ASCs retained their trilineage differentiation capacity after thawing, effectively differentiating into adipocytes, osteoblasts, and chondrocytes as demonstrated by positive staining with Oil Red O, Alizarin Red S, and Alcian Blue, respectively [4]. Colony-forming unit potential showed a trend toward higher clonogenicity in HFB-cells, though this did not reach statistical significance. Both manufacturing methods yielded cell survival rates exceeding 90% post-freeze-thaw, with TCP cells demonstrating slightly greater robustness [4]. Growth kinetics revealed no significant differences between the two cell types, with both showing similar proliferation patterns until reaching overconfluent conditions [4].
Table 2: Comparative Post-Thaw Characteristics of TCP vs. Bioreactor-Expanded MSCs
| Parameter | TCP-Expanded MSCs | Bioreactor-Expanded MSCs | Significance |
|---|---|---|---|
| Viability Post-Thaw | >90% | >90% | Comparable |
| CD73/CD90 Expression | >95% | >95% | Comparable |
| CD105 Expression | 75% (decreased post-thaw) | >95% (maintained) | Significant difference |
| CD274 Expression | Comparable pre/post-thaw | Increased by ~48% post-thaw | Difference balanced post-thaw |
| Trilineage Differentiation | Maintained | Maintained | Comparable |
| Colony-Forming Unit Potential | Moderate | Higher trend | Not significant |
| Proliferation Kinetics | No significant difference | No significant difference | Comparable |
| Wound Healing Assay (Fibroblast Migration) | Positive effect | Positive effect | Comparable |
Subpopulation Heterogeneity
A particularly insightful finding from the comparative study concerns the differential impact of expansion systems on MSC heterogeneity. The research identified distinct immunophenotypical subpopulations based on co-expression patterns of surface markers, revealing that TCP and HFB cultures support different subpopulations, influencing heterogeneity within ASC cultures [4]. After thawing, TCP-expanded cells became less variable while HFB-expanded cells became more variable during the freeze-thaw process [4]. Specifically, for subpopulation SPA2 (defined as CD73+, CD90+, CD105+), a significant difference was observed between expansion systems before freezing, which became even larger after thawing as SPA2 significantly decreased in TCP cells [4]. These findings highlight that while core MSC functionality is preserved regardless of expansion platform, the freeze-thaw process drives differential changes in subpopulations between systems, potentially influencing long-term performance in specific therapeutic applications.
Detailed Experimental Protocols
To enable replication of the key comparative studies cited in this guide, this section provides detailed methodologies for the critical experiments evaluating post-thaw MSC characteristics.
Expansion and Cryopreservation Protocol
The comparative study between TCP and HFB systems employed a carefully designed expansion protocol to ensure comparable population doublings between platforms despite their different passaging schedules [4]. For HFB expansion, one-fifth of ASCs were seeded in a hollow fiber bioreactor system (1.7 m²) for a single passage. For equivalent TCP expansion, fourth-fifth ASCs (equivalent to a quarter of HFB-cells) were seeded into a single T175 TCP flask (0.175 m²) and expanded 1:3 until passage 4, theoretically yielding 27 T175 flasks (totaling 0.47 m²) [4]. For practical considerations, only one-third of TCP cells were continued in each subsequent passage. HFB cells at P1 and TCP cells at P4 were cryopreserved using the slow freezing method, which remains the recommended technique for clinical and laboratory MSC cryopreservation due to its operational simplicity and minimal contamination risk [21]. The cryopreservation process involved mixing cells with cryoprotective agents (typically containing 5%-10% DMSO), gradual cooling to -80°C, followed by long-term storage in liquid nitrogen at -196°C [19] [21]. Thawing was performed by rapidly warming cryovials in a 37°C water bath until ice crystals were dissolved, followed by centrifugation to remove cryoprotective agents [21].
Post-Thaw Characterization Methods
Comprehensive post-thaw analysis included immunophenotyping, functional assays, and potency evaluations. Surface marker expression was analyzed using flow cytometry panels assessing both classic MSC markers (CD73, CD90, CD105) and additional markers (CD29, CD201, CD36, CD31, Stro-1, CD166, CD200, CD248, CD271, CD146, CD34, CD274) to identify immunophenotypic changes and subpopulations [4]. Trilineage differentiation capacity was evaluated by inducing adipogenic, osteogenic, and chondrogenic differentiation in culture, with successful differentiation confirmed by positive staining with Oil Red O (adipocytes), Alizarin Red S (osteoblasts), and Alcian Blue (chondrocytes), respectively [4]. Control cells maintained in complete medium without differentiation inducers showed no such staining. Colony-forming unit (CFU) assays were performed to assess clonogenicity by seeding cells at low density and counting formed colonies after appropriate incubation periods [4]. Proliferation potential was determined through growth kinetics studies, monitoring cell numbers over time with observation of detachment in overconfluent conditions due to excessive crowding, waste accumulation, or nutrient depletion [4]. Biological activity relevant to therapeutic applications was assessed through wound healing scratch assays, evaluating the influence of ASC paracrine effects on human dermal fibroblast (HDF) migration, along with proliferation assays to examine effects on HDF growth [4].
Diagram Title: Post-Thaw MSC Characterization Framework
The Scientist's Toolkit: Essential Reagents and Materials
Successful comparison of expansion platforms requires specific reagents and materials designed to maintain MSC properties throughout expansion, cryopreservation, and post-thaw evaluation. The following toolkit compiles essential solutions and their functions based on current comparative studies.
Table 3: Essential Research Reagents for MSC Expansion and Cryopreservation Studies
| Reagent/Material | Function/Purpose | Examples/Formulations |
|---|---|---|
| Xeno-Free Culture Medium | Supports clinical-grade MSC expansion without animal components | Stemline XF MSC medium [18] |
| Cryopreservation Solutions | Protects cells during freeze-thaw process; minimizes cryo-injury | NutriFreez (10% DMSO), CryoStor CS5/CS10, Plasmalyte-A with 5% HA and 10% DMSO [19] |
| Cryoprotective Agents (CPAs) | Prevents ice crystal formation; reduces osmotic stress during freezing | DMSO, sucrose, trehalose, ethylene glycol [21] |
| Microcarriers | Provides surface for adherent cell growth in suspension bioreactors | Gelatin methacrylate, polystyrene, alginate microcarriers [18] |
| Dissociation Reagents | Detaches adherent cells from substrates for passaging and harvesting | Enzymatic treatments (trypsin/accutase) with mechanical stress [18] |
| Flow Cytometry Antibodies | Characterizes immunophenotype and identifies subpopulations | CD73, CD90, CD105, CD45, CD34, CD14, CD19, HLA-DR [4] [17] |
| Differentiation Kits/Reagents | Confirms trilineage differentiation potential | Adipogenic: Oil Red O; Osteogenic: Alizarin Red S; Chondrogenic: Alcian Blue [4] |
The comparative analysis of TCP and bioreactor expansion systems reveals a nuanced landscape for MSC manufacturing. TCP systems remain valuable for research-scale applications where cost-effectiveness and flexibility are prioritized, while bioreactor systems offer clear advantages for clinical-scale production requiring standardization and scalability [4]. Critically, both expansion methods can produce functionally competent MSCs after cryopreservation, maintaining viability, differentiation capacity, and therapeutic potential in wound healing assays [4]. However, the observed differences in surface marker expression (particularly CD105) and subpopulation dynamics highlight that the expansion platform choice introduces distinct biological signatures that persist through cryopreservation [4].
For researchers and therapy developers, these findings suggest a context-dependent selection process. Early-stage research and proof-of-concept studies may benefit from the accessibility of TCP systems, while clinical translation efforts should prioritize bioreactor platforms to ensure manufacturing robustness and regulatory compliance. Future research directions should focus on optimizing cryopreservation protocols specifically tailored to each expansion system, further exploring the functional implications of the observed phenotypic differences, and developing advanced bioreactor technologies that better preserve MSC heterogeneity and potency throughout the manufacturing pipeline. As MSC therapies continue to advance toward widespread clinical application, understanding these platform-dependent characteristics becomes increasingly essential for developing effective, reproducible cell-based therapeutics.
Why Cryopreservation is a Critical Bottleneck in Clinical Translation
The successful clinical translation of cell-based therapies, particularly those utilizing Mesenchymal Stem/Stromal Cells (MSCs), hinges on the ability to reliably preserve, store, and transport living cellular products. Cryopreservation promises to break logistical barriers by enabling long-term storage and creating "off-the-shelf" availability for cell therapies [22] [23]. However, the process itself presents a critical bottleneck that can compromise both product quality and therapeutic efficacy. Conventional cryopreservation methods, largely unchanged since the 1950s, often prove suboptimal for clinical applications where minor alterations in post-thaw cell viability, phenotype, or function can significantly impact patient outcomes [23].
This bottleneck becomes particularly pronounced when considering the expansion systems used to produce therapeutic cells. Traditional two-dimensional tissue culture polystyrene (TCP) flasks and advanced three-dimensional bioreactor systems represent fundamentally different manufacturing paradigms, each imparting distinct characteristics to the cells they produce. Understanding how cells from these different expansion systems withstand the rigors of cryopreservation is essential for developing robust clinical-grade cell therapies. This article examines the experimental evidence comparing TCP versus bioreactor-expanded MSCs after cryopreservation, providing researchers with critical data to inform their therapeutic development strategies.
Fundamental Challenges in Cryopreservation
Physical and Chemical Stresses During Freeze-Thaw
Cryopreservation imposes multiple stressors on cellular systems, including solute concentration effects, ice crystal formation, membrane damage, and oxidative stress [22]. During freezing, as water transforms to ice, remaining solutes become concentrated in a potentially toxic hypertonic solution. Intracellular ice crystals can physically disrupt organelles and membrane structures. The thawing process presents equal challenges, particularly the risk of devitrification (ice crystal formation during warming) if the critical warming rate is not achieved [24].
The Scalability Challenge
A particularly significant bottleneck emerges during scale-up. While small-volume samples (typically 0.2 mL) can be successfully vitrified and rewarmed using conventional methods, larger volumes routinely used in clinical manufacturing face substantial technical hurdles. Table 1 summarizes the critical limitations observed during scale-up of cryopreservation protocols.
Table 1: Impact of Sample Volume on Cryopreservation Outcomes
| Sample Volume | Cooling Rate (°C/min) | Warming Rate (°C/min) | Devitrification Observed | Cell Viability Post-Thaw |
|---|---|---|---|---|
| 0.2-1 mL (Gold Standard) | >50°C/min | >50°C/min | No | 77.5 ± 9.6% |
| 8 mL | ~49.5°C/min | ~48.1°C/min | Slight | Moderate decrease |
| 20 mL | ~19.8°C/min | ~16.7°C/min | Yes | 38.5 ± 2.9% |
| 30 mL | ~4.9°C/min | ~4.5°C/min | Yes | ~40% |
Convective warming in water baths creates significant temperature gradients in larger volumes, with the center of samples warming much slower than the edges. This non-uniform rewarming causes devitrification and ice crystal formation, dramatically reducing cell viability [24]. This scalability challenge represents a fundamental bottleneck in producing clinically relevant cell quantities.
Comparative Analysis: TCP vs. Bioreactor-Expanded MSCs
Experimental Models and Culture Systems
Research directly comparing TCP and bioreactor-expanded MSCs after cryopreservation requires careful experimental design to ensure meaningful comparisons. One comprehensive study established equivalent population doublings between a hollow fiber bioreactor (HFB) system and conventional TCP flasks [4]. The HFB system (1.7 m² surface area) cultured cells for a single passage, while TCP-expanded cells (seeded at one-quarter the density of HFB cells) underwent expansion through multiple passages (to P4) to achieve equivalent expansion [4].
Table 2: Culture System Parameters for Comparative Studies
| Parameter | Tissue Culture Polystyrene (TCP) | Hollow Fiber Bioreactor (HFB) |
|---|---|---|
| Surface Area | 0.175 m² (T175 flask) | 1.7 m² |
| Passage Schedule | Multiple passages (to P4) | Single passage |
| Labor Intensity | High (manual handling) | Low (automated system) |
| Scalability | Limited | High |
| Consistency | Variable between operators | Highly reproducible |
| Initial Cost | Low | High |
Other bioreactor systems used in MSC expansion include spinner flasks, perfusion bioreactors, and rotating wall vessels [25]. The biaxial rotating (BXR) bioreactor has demonstrated particular promise, achieving superior cellularity and more homogeneous cell distribution in large (785 mm³) macroporous scaffolds compared to other systems [25].
Post-Thaw Viability and Phenotypic Stability
The freeze-thaw process differentially affects TCP and bioreactor-expanded cells, with notable impacts on both viability and surface marker expression. While both systems typically yield post-thaw viability exceeding 90%, TCP-expanded cells often demonstrate greater robustness in maintaining this viability [4].
More significantly, cryopreservation drives distinct immunophenotypic changes depending on the expansion system. Table 3 summarizes key differences in surface marker expression after cryopreservation.
Table 3: Phenotypic Changes After Cryopreservation of TCP vs. Bioreactor-Expanded MSCs
| Surface Marker | TCP-Expanded MSCs (Post-Thaw) | Bioreactor-Expanded MSCs (Post-Thaw) | Functional Significance |
|---|---|---|---|
| CD105 | Significant decrease (to ~75% positive) [4] | Maintained high expression | TGF-β receptor, associated with multipotency |
| CD73, CD90 | Maintained high expression (>95%) [4] | Maintained high expression (>95%) | Standard MSC markers |
| CD274 (PD-L1) | Significant increase (∼48%) [4] | Increased to comparable levels with TCP | Immunomodulatory protein |
| CD29, CD201 | Maintained high expression (~100%) | Maintained high expression (~100%) | Adhesion and progenitor markers |
| CD34, CD45 | Maintained low expression | Maintained low expression | Hematopoietic markers (negative) |
The significant decrease in CD105 expression in TCP-expanded cells after thawing is particularly noteworthy, as this transforming growth factor-beta receptor is associated with MSC multipotency [4]. This suggests that cryopreservation may more substantially impact the differentiation potential of TCP-expanded cells compared to their bioreactor-expanded counterparts.
Functional Characteristics Post-Thaw
Despite phenotypic differences, functional assessments reveal important insights about post-thaw performance:
- Trilineage Differentiation: Both TCP and bioreactor-expanded MSCs maintain adipogenic, osteogenic, and chondrogenic differentiation capacity after cryopreservation [4].
- Proliferation Potential: No significant differences in growth kinetics are typically observed between systems after thawing [4].
- Clonogenicity: While not reaching statistical significance in all studies, bioreactor-expanded cells may demonstrate higher colony-forming potential post-thaw [4].
- Therapeutic Function: Paracrine functions critical to MSC therapeutic mechanisms, including effects on fibroblast migration in wound healing models, appear preserved regardless of expansion system [4].
Interestingly, one study demonstrated that cryopreservation of MSCs directly on β-TCP scaffolds actually enhanced their osteogenic potential compared to non-frozen controls, with increased expression of early osteogenic markers (RunX2, Col1, ALPL) [26]. This suggests that the cryopreservation process itself may activate specific cellular pathways that influence subsequent differentiation behavior.
Molecular Insights: Signaling Pathways Affected by Cryopreservation
Cellular Stress Response Pathways
The freeze-thaw process activates specific molecular pathways that differ between culture systems. Research indicates that cryopreservation influences:
- Hypoxia-Related Pathways: Cryopreservation upregulates HIF1α expression and its target genes (PDK1, SLC2A1, EGLN1, BNIP3), potentially mimicking hypoxic conditions that may enhance certain MSC functions [26].
- Cold Shock Response: The cold shock protein YBX1 shows significantly enhanced expression following cryopreservation, remaining elevated for at least 7 days post-thaw [26]. YBX1 functions as an important regulator of transcription and may influence MSC phenotype recovery.
- Metabolic Reprogramming: Changes in PDK1 expression suggest a shift toward glycolysis, potentially supporting post-thaw recovery by reducing oxidative stress [26].
Cryopreservation activates cellular stress response pathways that influence post-thaw MSC function. The diagram illustrates how freezing stressors trigger molecular pathways that ultimately affect functional outcomes, potentially differing between TCP and bioreactor-expanded cells.
Impact on Therapeutic Mechanisms
MSCs exert their therapeutic effects primarily through paracrine signaling and immunomodulation rather than direct engraftment and differentiation [17]. Key mechanisms include:
- Immunomodulation: Through secretion of PGE2, IDO, and PD-L1, MSCs inhibit T-cell proliferation and polarize macrophages toward anti-inflammatory M2 phenotypes [17].
- Trophic Factor Secretion: VEGF, bFGF, HGF, and IGF-1 promote angiogenesis, reduce fibrosis, and inhibit cell death [17].
- Mitochondrial Transfer: A recently discovered mechanism where MSCs donate mitochondria to injured cells via tunneling nanotubes, restoring bioenergetic function in conditions like ARDS and myocardial ischemia [17].
The preservation of these functions after cryopreservation is essential for therapeutic efficacy. While direct comparisons between TCP and bioreactor-expanded cells are limited, evidence suggests that 3D culture systems may better maintain these functions post-thaw due to their more physiological culture environment.
Innovative Approaches to Overcome the Bottleneck
Advanced Cryopreservation Technologies
Several innovative approaches are emerging to address the cryopreservation bottleneck:
- Nanowarming Technology: This approach uses magnetic nanoparticles (typically 10nm magnetite) dispersed in cryoprotectant solutions that generate heat uniformly when exposed to alternating magnetic fields. This enables rapid, uniform rewarming of large volumes (up to 20mL demonstrated), overcoming devitrification problems associated with conventional water bath rewarming [24].
- DMSO-Free Cryoprotectants: Alternatives to conventional DMSO-based cryoprotectants are emerging, such as CryoOx, which demonstrate comparable viability while eliminating DMSO toxicity concerns [27]. StemCell Keep, a DMSO-free solution containing carboxylated ε-poly-l-lysine, has shown success with pluripotent stem cells [24].
- Biomimetic Approaches: Learning from natural organisms that tolerate extreme environmental stressors provides templates for improved cryopreservation strategies, including ice-binding proteins and stress response modulators [23].
Integrated Bioprocessing Strategies
The most promising approaches integrate expansion and preservation strategies:
- Cryopreservation on Scaffolds: Direct cryopreservation of MSCs on β-TCP scaffolds maintains cell viability and distribution while simplifying logistics for bone tissue engineering applications [26].
- Controlled Ice Nucleation: Precise control of ice nucleation temperatures improves consistency and reduces cryoinjury across large cell batches [22].
- Quality-by-Design Frameworks: Implementing QbD principles helps identify critical process parameters (CPPs) and critical quality attributes (CQAs) for more robust cryopreservation protocols [20].
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 4: Key Reagents and Solutions for Cryopreservation Research
| Reagent/Solution | Function | Example Products/Formulations |
|---|---|---|
| DMSO-Based Cryomedium | Traditional cryoprotectant | Commercial DMSO solutions (e.g., CryoStor) |
| DMSO-Free Alternatives | Cryoprotection without DMSO toxicity | CryoOx, StemCell Keep |
| Magnetic Nanoparticles | Enable nanowarming technology | 10nm magnetite nanoparticles |
| Viability Assays | Assess post-thaw cell health | Flow cytometry with Annexin V/PI, Calcein AM |
| Immunophenotyping Panels | Characterize surface marker expression | Antibodies against CD73, CD90, CD105, CD274 |
| Differentiation Kits | Verify trilineage potential post-thaw | Osteogenic, adipogenic, chondrogenic media |
| Cryocontainers | Cell storage during freezing | Cryobags, cryovials (1-30mL capacities) |
Experimental workflow for comparing TCP vs. bioreactor-expanded MSCs after cryopreservation. The diagram outlines key steps from cell expansion through post-thaw assessment, highlighting differential outcomes like CD105 decrease in TCP-expanded cells.
Cryopreservation remains a critical bottleneck in the clinical translation of MSC therapies, with the expansion system significantly influencing post-thaw outcomes. TCP and bioreactor expansion systems produce cells with distinct phenotypic and functional characteristics that respond differently to cryopreservation stresses. While bioreactor systems offer advantages in scalability and consistency, both systems can yield clinically viable cells with appropriate protocol optimization.
The emerging understanding of molecular responses to cryopreservation, including HIF1α and YBX1 activation, provides new opportunities for targeted intervention. Combined with innovative technologies like nanowarming and DMSO-free cryoprotectants, these insights promise to overcome current limitations. For researchers developing MSC-based therapies, systematic evaluation of cryopreservation impacts within their specific manufacturing workflow is essential—what works for TCP-expanded cells may not optimize outcomes for bioreactor-expanded products. Through continued refinement of integrated expansion and preservation strategies, the field can overcome the cryopreservation bottleneck and realize the full clinical potential of MSC therapies.
Scalable Manufacturing: From Flask-Based Culture to Automated Bioreactor Systems
In the context of advanced therapy medicinal product (ATMP) development, the "expansion" of biological products is a critical manufacturing step. This guide explores two distinct interpretations of "expansion": the scaling of Transmission Control Protocols in networking and the scaling of Mesenchymal Stromal/Stem Cells in biomanufacturing. While seemingly disparate fields, both face core challenges of achieving scalability, maintaining quality, and managing operational intensity. For MSC therapies, expansion refers to the process of increasing cell numbers from a small starting sample to clinically relevant quantities, often numbering in the hundreds of millions or billions of cells [20]. The conventional methods for this expansion, primarily two-dimensional (2D) tissue culture flasks, present significant limitations in scalability, reproducibility, and labor requirements, much like how conventional TCP faces limitations in modern network environments [28] [4]. This guide objectively compares the performance of conventional TCP expansion protocols against emerging alternatives and draws parallels to conventional 2D flask-based MSC expansion versus bioreactor-based systems, providing researchers with critical experimental data and comparative frameworks for evaluation.
Conventional TCP Expansion Protocols
Foundational Protocol Characteristics
The Transmission Control Protocol (TCP) forms the backbone of reliable internet communication, employing a connection-oriented design that ensures ordered, error-checked data delivery. Conventional TCP expansion refers to efforts to enhance TCP's capabilities within its original architectural constraints, primarily through the addition of options within the limited header space [29]. The standard TCP header provides only 40 bytes of option space beyond the basic 20-byte header, creating a significant constraint for advanced functionality [29]. This limitation becomes particularly problematic when attempting to combine multiple sophisticated options such as Multipath TCP (MP-TCP), TCP Authentication Option (TCP-AO), and Selective Acknowledgement (SACK) in a single connection [29]. These expansion efforts aim to enhance TCP's capabilities for modern networking needs without fundamentally altering its core architecture, much like how 2D flask expansion attempts to scale MSC manufacturing within traditional laboratory constraints.
Key Conventional TCP Extensions
Multipath TCP (MP-TCP): enables a single TCP connection to use multiple paths simultaneously, improving throughput and resiliency. MP-TCP is implemented in Apple iOS and Mac OS X and several server load balancers, allowing connections to leverage multiple network interfaces without application changes [30].
TCP Extended Data Offset (EDO): an experimental protocol addressing TCP's option space limitation by extending the header capacity for all segments except the initial SYN. EDO allows combinations of advanced options like TCP-AO, Timestamps, and MP-TCP that would otherwise exceed the standard 60-byte header limit [29].
Data Center TCP (DCTCP): optimizes congestion notification for data center environments by providing more precise feedback on congestion levels. DCTCP has been implemented by Microsoft in Windows Server 2012 and Cisco in Nexus switches, improving throughput in high-performance computing environments [30].
HTTP/2 and QUIC: represent application-layer expansion approaches. HTTP/2 introduces multiplexing, header compression, and server push over traditional TCP [30], while QUIC Internet Connections implements TCP-like reliability over UDP, reducing connection establishment latency [28] [30].
Performance Limitations of Conventional TCP Expansion
Table 1: Performance Characteristics of TCP Extension Protocols
| Protocol | Latency Impact | Throughput Enhancement | Implementation Complexity | Middlebox Compatibility |
|---|---|---|---|---|
| MP-TCP | Moderate increase due to path management | High (utilizes multiple paths) | High (requires OS-level support) | Moderate (some middleboxes may strip options) |
| TCP EDO | Minimal increase | Moderate (enables better option combinations) | Moderate (kernel modifications required) | Low (experimental, limited deployment) |
| DCTCP | Decreased in data center environments | High in controlled environments | High (requires switch support) | Low (primarily for data centers) |
| QUIC | Significant decrease (0-RTT handshake) | High (avoids TCP head-of-line blocking) | High (user-space implementation) | High (uses UDP on port 443) |
| HTTP/2 | Decreased (multiplexing, header compression) | Moderate (better connection utilization) | Low (application layer only) | High (works over standard TCP) |
The experimental data for TCP EDO indicates it successfully enables combination of previously incompatible option sets, with testing showing the protocol can support header sizes up to 255 words (1020 bytes), dramatically expanding capacity beyond the conventional 60-byte limit [29]. However, this expansion comes with operational burdens, including implementation complexity and potential middlebox interference. Similarly, QUIC demonstrates measurable performance improvements, reducing connection establishment latency from 1-3 round trips in conventional TCP to 0-1 round trips, at the cost of significant protocol complexity and the challenge of operating over UDP [28] [30].
Labor Intensity and Implementation Challenges
Operational Complexities in TCP Expansion
Expanding TCP capabilities introduces significant operational burdens that mirror the labor intensity of manual MSC expansion. Protocol development and standardization for TCP extensions typically follow Internet Engineering Task Force processes spanning years, with EDO and related SYN extension options still in draft status as of 2025 [29] [31]. Implementation requires deep expertise in network stack programming and kernel development, as these modifications touch core networking functionality. Deployment challenges include network middlebox interference, where Network Address Translation devices, firewalls, and other intermediary systems may strip, modify, or block packets with unfamiliar TCP options [29]. Testing and validation present additional labor burdens, requiring sophisticated network testbeds and interoperability testing across diverse implementations. These challenges collectively contribute to the slow adoption of TCP extensions despite their technical benefits, reminiscent of how manual processes hinder scalable MSC manufacturing.
Maintenance and Monitoring Overheads
Maintaining expanded TCP implementations requires continuous monitoring and adjustment. Congestion Control algorithms like DCTCP need careful parameter tuning for specific network environments [30]. Path Management in MP-TCP demands monitoring of multiple network paths with potentially varying characteristics [30]. Security Considerations expand with each new option, requiring ongoing vulnerability assessment and patching [29]. Performance Monitoring must track non-standard metrics beyond conventional throughput and latency measurements. These maintenance activities demand specialized networking expertise and dedicated operational resources, creating significant labor overhead that must be factored into deployment decisions.
Conventional MSC Expansion in Biomanufacturing
2D Flask-Based Expansion Systems
Conventional MSC expansion relies primarily on two-dimensional tissue culture polystyrene flasks and multi-layer vessels. These systems involve labor-intensive manual processes including passaging, media changes, and cell harvesting [4]. The scalability of 2D systems is physically constrained by available surface area, requiring exponentially more incubator space and handling time as scale increases [20] [4]. This manual, open-process approach also introduces significant opportunities for contamination and operator-dependent variability, challenging manufacturing consistency [4]. Like conventional TCP facing option space limitations, 2D culture systems fundamentally limit MSC manufacturing capacity while requiring substantial manual intervention.
Bioreactor-Based Expansion Systems
Bioreactor systems address 2D limitations by enabling three-dimensional culture in controlled, automated environments. Multiple bioreactor technologies have emerged for MSC expansion, each with distinct characteristics. The Quantum Cell Expansion System is a hollow fiber bioreactor providing a closed, Good Manufacturing Practice-compliant platform for adherent cell culture [5]. Stirred-tank Bioreactors suspend cells with microcarriers providing attachment surfaces, enabling volumetric scaling [20]. Hollow Fiber Bioreactors like those evaluated in comparative studies provide high surface-to-volume ratios in cartridge formats [4]. These automated systems reduce manual labor while improving scalability and process control, analogous to how modern transport protocols automate connection optimization in networking.
Table 2: Comparative Analysis of MSC Expansion Systems
| Expansion System | Max Cell Yield | Labor Intensity | Process Control | Cryopreservation Outcomes | Scalability |
|---|---|---|---|---|---|
| Tissue Culture Flasks (2D) | Limited by surface area | High (manual operations) | Low (operator dependent) | Significant CD105 loss post-thaw [4] | Limited (linear with surface area) |
| Hollow Fiber Bioreactor | High (1.7m² surface area) | Low (automated feeding) | High (parameter monitoring) | Maintained CD105 expression [4] | Moderate (cartridge-based) |
| Stirred-Tank Bioreactor | Very high (volumetric scaling) | Moderate (setup then automated) | High (pH, DO, temperature control) | Maintained viability & differentiation [20] | High (litre-scale volumes) |
| Bio-Block Hydrogel | High (3D structure) | Low (minimal intervention) | Moderate (environment control) | Preserved secretome function [14] | Modular (puzzle-piece design) |
Experimental Comparison: Performance After Cryopreservation
Methodology for Comparative Analysis
The critical comparison between expansion systems evaluates MSC quality after cryopreservation, essential for off-the-shelf therapy availability. Experimental protocols follow standardized processes: cells are expanded in parallel systems, cryopreserved using controlled-rate freezers, then thawed and assessed. Key quality metrics include Viability Measurement via trypan blue exclusion [4], Immunophenotype Analysis by flow cytometry for CD73, CD90, CD105 markers [4], Trilineage Differentiation potential into adipogenic, osteogenic, and chondrogenic lineages [4] [14], Colony-Forming Unit assays for stemness quantification [4], and Secretome Analysis including extracellular vesicle production and function [14]. These standardized methodologies enable objective comparison between expansion platforms, revealing critical differences in how cells withstand cryopreservation stress.
Experimental Results and Performance Data
Recent studies demonstrate significant functional differences between MSC expansion methods post-cryopreservation. Research comparing hollow fiber bioreactors to tissue culture flasks showed notable CD105 expression retention in bioreactor-expanded cells (maintaining >95% expression) versus significant CD105 loss in flask-expanded cells (decreasing to ~75% positive) [4]. Both systems maintained high viability (>90%) post-thaw, but the phenotypic changes in flask-expanded cells suggest potential functional impacts [4]. In comparative studies of 3D culture systems, Bio-Block platforms demonstrated ~2-fold higher proliferation than spheroid and Matrigel systems with senescence reduced 30-37% and apoptosis decreased 2-3-fold [14]. Perhaps most significantly, secretome analysis revealed dramatic differences, with EV production increasing ~44% in Bio-Blocks while declining 30-70% in other systems [14]. These functional differences after cryopreservation highlight how expansion methodology influences critical therapeutic attributes.
Diagram 1: Experimental workflow comparing 2D vs. 3D expansion outcomes post-cryopreservation
Technological Convergence and Future Directions
Automated and Closed-System Solutions
Both TCP expansion and MSC biomanufacturing show convergent evolution toward automated, closed-system solutions that reduce labor intensity while improving performance. In networking, protocols like QUIC implement transport services at the application layer, bypassing operating system dependencies and middlebox interference [30]. Similarly, closed-system bioreactors like the Quantum system provide automated, GMP-compliant expansion platforms that minimize manual operations and contamination risk [5]. These systems incorporate advanced monitoring and control capabilities, with bioreactors tracking parameters like dissolved oxygen, pH, and metabolite concentrations [20], while network protocols implement sophisticated congestion control and path management [30]. The convergence toward integrated, purpose-built systems in both fields represents a recognition that incremental expansion of legacy systems faces fundamental limitations.
Standardization and Quality-by-Design Approaches
Future development in both fields emphasizes standardization and proactive quality management. The Quality-by-Design framework, as outlined in ICH Q8 guidelines, is being applied to MSC manufacturing to define Critical Quality Attributes and Critical Process Parameters early in development [20]. Similarly, TCP extension protocols are incorporating more rigorous testing requirements and implementation guidelines [29] [31]. For MSC expansion, this means identifying key quality attributes like immunophenotype, differentiation potential, and secretome function early, then designing processes to consistently achieve them [20] [32]. This systematic approach contrasts with the empirical, incremental development that characterized early expansion efforts in both fields, promising more robust and reproducible outcomes.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Platforms for Expansion Studies
| Reagent/Platform | Function | Application Notes |
|---|---|---|
| RoosterNourish MSC-XF Medium | Xeno-free expansion medium | Supports MSC growth without animal components [14] |
| PRIME-XV MSC Expansion XSFM | Serum-free medium formulation | Maintains stemness during expansion [32] |
| PRIME-XV FreezIS DMSO-Free | Cryopreservation without DMSO | Avoids DMSO toxicity while maintaining recovery [32] |
| Quantum Cell Expansion System | Hollow fiber bioreactor platform | Closed-system, GMP-compliant expansion [5] |
| Trypsin/EDTA Solution | Cell dissociation reagent | Standardized detachment from microcarriers or flasks [14] |
| Flow Cytometry Antibodies | Immunophenotype analysis | CD73, CD90, CD105 for ISCT criteria verification [4] |
| Trilineage Differentiation Kits | Functional potency assessment | Adipogenic, osteogenic, chondrogenic induction [4] [14] |
| AlamarBlue/MTT Assays | Viability and proliferation metrics | Quantitative growth kinetics measurement [4] |
Diagram 2: MSC expansion workflow from source material to quality verified product
The comparative analysis reveals fundamental trade-offs between conventional and advanced expansion methodologies in both networking and biomanufacturing domains. Conventional TCP expansion and 2D MSC culture share common limitations in scalability, labor requirements, and performance constraints. Emerging solutions in both fields address these limitations through architectural innovations: QUIC reimagines transport protocol design by building on UDP [30], while bioreactor systems transform MSC manufacturing through automation and environmental control [20] [5]. The experimental data demonstrates that expansion methodology significantly influences post-cryopreservation outcomes, with bioreactor-expanded MSCs better maintaining critical quality attributes like CD105 expression and secretome function [4] [14]. For researchers and therapy developers, selection criteria should prioritize scalability, labor efficiency, and quality consistency, favoring advanced expansion platforms that address the fundamental limitations of conventional approaches while providing a path to clinical and commercial manufacturing scale.
The selection of an appropriate bioreactor system is a critical decision in bioprocess development, with profound implications on cell yield, product quality, and process economics. This guide provides an objective comparison of three predominant technologies—hollow fiber bioreactors (HFB), stirred-tank bioreactors (STR), and microcarrier-based systems—framed within a research context examining tissue culture polystyrene (TCP) versus bioreactor-expanded mesenchymal stromal/stem cells (MSCs) after cryopreservation. For researchers and drug development professionals, understanding the performance characteristics, scalability, and post-preservation outcomes of each system is essential for advancing cell-based therapies from bench to bedside. We synthesize experimental data across multiple studies to highlight the comparative advantages and limitations of each platform, with particular attention to their impact on MSC characteristics following cryopreservation, a crucial consideration for developing "off-the-shelf" cell therapies.
Fundamental Operating Principles
Hollow Fiber Bioreactors (HFB): These systems utilize a network of semi-permeable hollow fibers to create a high-surface-area environment for cell culture. Nutrients and gases diffuse across the fiber membranes, providing efficient exchange while protecting cells from shear stress. Cells typically reside in the extracapillary space, creating a tissue-like environment with high cell densities [33]. HFBs can be operated in various modes, including perfusion, which continuously supplies nutrients and removes waste products.
Stirred-Tank Bioreactors (STR): As workhorses of bioprocessing, STRs rely on mechanical impellers for mixing and oxygenation. They offer homogeneous culture conditions and are well-characterized for scale-up. Traditional STRs are suitable for suspension-adapted cells but can generate significant shear stress, potentially damaging sensitive cell types [33]. Modern STRs often incorporate design modifications to mitigate shear effects.
Microcarrier-Based Systems: These platforms bridge the gap between adherent culture and suspension systems by providing small beads (microcarriers) with high surface-to-volume ratios as attachment substrates for anchorage-dependent cells. They are typically implemented in stirred-tank configurations, enabling scalable cultivation of cells that require surface attachment [34] [35].
Comparative Performance Analysis
The table below summarizes key performance metrics for hollow fiber, stirred-tank, and microcarrier-based bioreactor systems, synthesizing data from multiple experimental studies.
Table 1: Comprehensive Performance Comparison of Bioreactor Technologies
| Performance Parameter | Hollow Fiber Bioreactor | Stirred-Tank Bioreactor | Microcarrier-Based System |
|---|---|---|---|
| Max Cell Density | Very high (3D tissue-like densities) [33] | Moderate to high (depends on cell type) [34] | High (1.2-3.5 × 10^6 cells/mL for BHK-21) [34] |
| Shear Stress Impact | Low (protective environment) [33] | High (mechanical agitation) [33] | Moderate (depends on agitation) [7] |
| Scalability | Modular expansion [33] | Well-established scale-up [33] | Scalable but complex [35] |
| Mass Transfer Efficiency | High (direct diffusion) [33] [36] | Mixing-dependent [33] | Mixing and diffusion-dependent [35] |
| Downstream Processing | Simplified (concentrated product) [33] | Complex (cell separation required) [33] | Complex (cell detachment needed) [34] |
| Culture Volume Requirements | Low media consumption [33] | High media consumption [33] | Moderate to high media consumption [34] |
| Post-Thaw Viability (MSCs) | >90% [4] | Varies with cell type | Similar to STR (cell-dependent) [34] |
| CD105 Retention (Post-Thaw MSCs) | Maintained [4] | Significant decrease (75% positive) [4] | Cell-dependent |
| Relative Cost Structure | High initial investment, lower media costs [33] [4] | Lower initial investment, higher operational costs [33] [4] | Moderate investment, variable operational costs [34] |
Impact on MSC Characteristics Post-Cryopreservation
Experimental Framework for TCP vs. Bioreactor-Expanded MSCs
Understanding how expansion systems affect MSC properties after cryopreservation is crucial for clinical applications. A comparative study established equivalent population doublings between HFB and TCP systems: HFB cells at passage 1 were compared with TCP cells at passage 4 to ensure comparable replicative history before cryopreservation [4]. Cells were cryopreserved using standard protocols with cryoprotective agents, then analyzed post-thaw for phenotypic and functional characteristics.
Table 2: Post-Thaw Characteristics of MSCs Expanded in Different Systems
| Characteristic | HFB-Expanded MSCs | TCP-Expanded MSCs | Analytical Method |
|---|---|---|---|
| Viability | >90% [4] | >90% [4] | Flow cytometry |
| CD73/CD90 Expression | Maintained (>95%) [4] | Maintained (>95%) [4] | Flow cytometry |
| CD105 Expression | Maintained (>95%) [4] | Significant decrease (75% positive) [4] | Flow cytometry |
| CD274 (PD-L1) Expression | Increased post-thaw (comparable to TCP) [4] | High expression maintained [4] | Flow cytometry |
| Trilineage Differentiation | Preserved [4] | Preserved [4] | Oil Red O, Alizarin Red S, Alcian Blue staining |
| Colony-Forming Units | Trend toward higher potential [4] | Robust potential [4] | CFU assay |
| Subpopulation Heterogeneity | Increased variability post-thaw [4] | Decreased variability post-thaw [4] | Cluster analysis of surface markers |
| Functional Effects on Fibroblasts | Maintained (migration promotion) [4] | Maintained (migration promotion) [4] | Scratch assay |
Signaling Pathways in MSC Mechanotransduction
Bioreactor culture environments exert mechanical forces that influence MSC biology through mechanotransduction pathways. The diagram below illustrates key signaling mechanisms triggered by different bioreactor environments and their impact on post-thaw MSC characteristics.
Diagram Title: Mechanotransduction Pathways in Bioreactor Culture Affecting Post-Thaw MSC Quality
Experimental Protocols for Comparative Studies
Microfluidic Bioreactor Protocol for Shear Stress Studies
The following detailed methodology was employed to investigate shear stress effects on MSC focal adhesions and cryopreservation outcomes, utilizing a customized microfluidic bioreactor [37]:
Device Preparation: Fabricate microfluidic cell observation chambers with appropriate surface treatment for cell adhesion. Sterilize the system before cell seeding.
Cell Seeding: Seed human MSCs (umbilical cord blood-derived) at appropriate density in MSC-specific growth medium. Allow initial attachment under static conditions.
Shear Stress Application: Connect the microchannel to a syringe pump system. Induce controlled shear stresses within the range of 0.1-0.4 dyn/cm² (equivalent to 0.002-0.004 μbar) by adjusting flow rates from 0.0009 m/s to 0.0015 m/s inlet velocity. Maintain shear conditions for 8 days.
Numerical Analysis: Perform computational fluid dynamics (CFD) modeling to validate shear stress distribution throughout the microchannel. Confirm consistent shear gradients at different channel depths.
Cryopreservation: After 8 days of shear exposure, harvest cells and cryopreserve using a controlled-rate freezer with cryoprotective agents (DMSO, trehalose, ectoine, or catalase).
Post-Thaw Analysis: Assess cell viability, focal adhesion points (vinculin expression via immunostaining), phenotype retention (flow cytometry for CD90, CD44, CD105, CD73), and functional characteristics.
Hollow Fiber vs TCP Expansion and Cryopreservation Protocol
This protocol details the comparative expansion of MSCs in hollow fiber bioreactors versus TCP flasks with subsequent cryopreservation analysis [4]:
Experimental Design: Establish equivalent population doublings by seeding one-fifth of ASCs in a HFB system (1.7 m²) for a single passage, while seeding four-fifths ASCs (equivalent to a quarter of HFB-cells) into T175 TCP flasks (0.175 m²) and expanding 1:3 until passage 4.
HFB Expansion: Culture adipose-derived stem cells (ASCs) in the hollow fiber bioreactor according to manufacturer specifications. Maintain appropriate perfusion rates and gas exchange.
TCP Expansion: Culture ASCs in parallel using standard tissue culture methods with sequential passaging at 70-80% confluence.
Cryopreservation: Cryopreserve HFB cells at P1 and TCP cells at P4 using standard freezing protocols with controlled cooling rates and cryoprotectant solutions.
Post-Thaw Analysis: Upon thawing, assess:
- Viability via flow cytometry
- Surface marker expression (CD73, CD90, CD105, CD29, CD201, CD36, CD31, Stro-1, CD166, CD200, CD248, CD271, CD146, CD34, CD274)
- Immunophenotypical subpopulations via cluster analysis
- Trilineage differentiation potential (adiopogenic, osteogenic, chondrogenic)
- Colony-forming unit capacity
- Proliferation kinetics
- Functional effects on fibroblast migration (scratch assay)
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Materials for Bioreactor MSC Studies
| Item | Function/Application | Example Specifications |
|---|---|---|
| Hollow Fiber Bioreactor | 3D cell expansion with high density | 1.7 m² surface area, appropriate molecular weight cutoff [4] |
| Microcarriers | Surface for adherent cell growth in suspension | Cytodex series (e.g., Cytodex 1, Cytodex 3) [34] [35] |
| Single-Use Bioreactor Systems | Scalable culture with reduced contamination risk | Ambr, Biostat (Sartorius); BioBLU (Eppendorf); Mobius (Merck) [38] |
| Cryoprotective Agents | Cell preservation during freezing | DMSO, trehalose, ectoine, catalase [37] |
| Surface Marker Antibodies | Phenotypic characterization | CD90, CD44, CD105, CD73, CD14, CD34, CD45 [37] [4] |
| Differentiation Media | Multilineage potential assessment | Adipogenic, osteogenic, chondrogenic induction cocktails [4] |
| Microfluidic Bioreactor | Shear stress studies | Custom-designed cell observation chamber with flow control [37] |
| Flow Cytometer | Immunophenotyping and viability analysis | Multi-parameter systems with appropriate laser configurations [37] [4] |
| Controlled-Rate Freezer | Reproducible cryopreservation | Programmable cooling rate capability (e.g., -1°C/min) [37] |
Comparative Workflow: MSC Expansion to Post-Thaw Analysis
The following diagram illustrates the comprehensive experimental workflow from cell expansion through cryopreservation and post-thaw analysis, highlighting critical decision points and comparative assessments across culture platforms.
Diagram Title: Experimental Workflow for Comparative MSC Expansion and Cryopreservation
This comparison guide demonstrates that each bioreactor technology offers distinct advantages for MSC expansion with significant implications for post-cryopreservation outcomes. Hollow fiber bioreactors provide a protective, high-density environment that maintains CD105 expression and reduces media consumption, but require substantial initial investment. Stirred-tank bioreactors offer well-established scalability but generate higher shear stress. Microcarrier systems enable scalable expansion of adherent cells but introduce downstream processing complexity.
Critically, the expansion system selectively influences MSC subpopulations and specific surface marker expression after cryopreservation, particularly affecting CD105 and CD274 retention. However, fundamental MSC functional characteristics—including trilineage differentiation potential and paracrine effects on fibroblast migration—remain preserved across platforms post-thaw.
For researchers developing MSC-based therapies, selection criteria should prioritize alignment with target product profiles: hollow fiber systems for high-density, shear-sensitive cultures with enhanced post-thaw phenotype retention; stirred-tank reactors for suspension-adapted cells at large scale; and microcarrier-based approaches for scalable expansion of adherence-dependent cells. These findings underscore the importance of matching bioreactor technology to both process needs and critical quality attributes of the final cryopreserved cell product.
The transition from manual, open-process cell culture in tissue culture polystyrene (TCP) flasks to automated closed-system bioprocessing represents a pivotal evolution in the manufacturing of advanced therapy medicinal products (ATMPs), including mesenchymal stromal cell (MSC)-based therapies [39]. These automated platforms address critical challenges in cell therapy manufacturing by reducing human error, minimizing contamination risks, improving process consistency, and enabling scalability while maintaining compliance with Good Manufacturing Practice (GMP) standards [40] [41]. For MSC-based therapies specifically, which often require large cell quantities (millions to hundreds of millions) for clinical efficacy, automated bioreactor systems offer a solution to the limitations of traditional expansion methods, which are labor-intensive, time-consuming, and limited in scale [39].
This guide provides an objective comparison of three leading automated closed-system platforms—Quantum (Terumo BCT), CliniMACS Prodigy (Miltenyi Biotec), and Cocoon (Lonza)—within the research context of comparing TCP versus bioreactor-expanded MSCs after cryopreservation. The analysis focuses on technical specifications, experimental performance data, and implementation requirements to inform researchers, scientists, and drug development professionals in their platform selection process.
The Quantum system employs a hollow fiber bioreactor technology, the CliniMACS Prodigy utilizes an integrated adherent cell culture process, and the Cocoon Platform provides a flexible, modular approach to automated cell therapy manufacturing [39] [42] [43]. Each system represents a distinct technological approach to addressing the challenges of scalable, GMP-compliant cell manufacturing.
Table 1: Technical Specifications of Automated Closed-System Platforms
| Feature | Quantum Cell Expansion System | CliniMACS Prodigy | Cocoon Platform |
|---|---|---|---|
| Manufacturer | Terumo BCT | Miltenyi Biotec | Lonza |
| Technology Type | Hollow fiber bioreactor | Integrated cell processing with Adherent Cell Culture (ACC) | Automated, single-use cassette-based system |
| System Footprint | Not specified | Not specified | Compact design for decentralized manufacturing |
| Process Integration | Cell expansion only | End-to-end from cell isolation to harvest | End-to-end from cell culture to formulation |
| Culture Surface Area | 21,000 cm² (equivalent to 120 T-175 flasks) [39] | Configurable with CellSTACK | Scalable across production phases |
| Culture Vessel | Single-use, coated hollow fiber bioreactor | Tubing set (TS730) with culture vessels | Single-use, sterile cassettes |
| Key Features | Continuous medium exchange; Gas connection for normoxic/hypoxic culture | Automated cell isolation, inoculation, cultivation, media change, and harvest | Integrated magnetic selection; User-friendly software; Highly customizable protocols |
| Supported Cell Types | Adherent cells (BM-MSCs, AT-MSCs, UC-MSCs) | Broad range (BM-MSCs, AT-MSCs, UC-MSCs, CAR-T cells) | Autologous and allogeneic therapies (CAR-T cells, convertibleCAR T cells) |
Comparative Performance Data for MSC Expansion
Experimental studies have demonstrated the capabilities of these platforms for expanding MSCs while maintaining critical quality attributes. The data reveals differences in yield, efficiency, and functional characteristics of the cells produced.
Expansion Yield and Efficiency
Table 2: Performance Metrics for MSC Expansion Across Platforms
| Performance Metric | Quantum System | CliniMACS Prodigy | Cocoon Platform | Traditional TCP Flasks |
|---|---|---|---|---|
| Reported MSC Yield | 100-276 × 10⁶ BM-MSCs in 7 days (from 20 × 10⁶ seed) [39] | 29-50 × 10⁶ equine MSCs at P0 [39] | >1 × 10⁹ CAR+ T cells (comparable MSC yields possible) [44] | Varies significantly with scale and manual handling |
| Process Duration | 7-day expansion demonstrated [39] | 10-day procedure demonstrated [39] | Varies with protocol | Weeks for clinical-scale yields |
| Manual Manipulation | Reduced from ~54,400 to 133 steps compared to flasks [39] | Fully automated from isolation to harvest | Fully automated, end-to-end | High (all steps manual) |
| Needed Passages | Reduced to half compared to flask-based propagation [39] | Not specified | Not specified | Multiple passages required |
| Cell Viability Post-Cryopreservation | Maintained | Not specified | >70% viability post-thaw for T cells [44] | >90% (TCP-expanded ASCs) [4] |
Functional Characteristics of Expanded MSCs
Research comparing hollow fiber bioreactor (HFB) systems like Quantum against traditional TCP flasks provides critical insights into how expansion methodology influences MSC characteristics, particularly after cryopreservation—a key requirement for creating "off-the-shelf" therapies [4].
A comprehensive study comparing cryopreserved adipose-derived stem cells (ASCs) expanded in HFB versus TCP systems found that both methods produced fully functional ASCs post-thaw, though with some notable differences in immunophenotype [4]:
- Immunophenotype Stability: Both systems maintained high expression (>95%) of characteristic MSC markers CD73 and CD90 before and after cryopreservation. However, TCP-expanded cells showed a significant decrease in CD105 expression after freeze-thawing (from >95% to 75%), while HFB-expanded cells maintained stable CD105 expression [4].
- Subpopulation Dynamics: The two expansion methods supported different MSC subpopulations, influencing heterogeneity. TCP cultures showed a significant increase in CD73+/CD90+/CD105- subpopulations after thawing, while HFB cultures maintained more stable subpopulation distributions [4].
- Functional Capacity: Despite immunophenotypic differences, both expansion methods yielded cells with equivalent differentiation capacity (adirogenic, osteogenic, chondrogenic), colony-forming potential, proliferation kinetics, and effects on fibroblast migration in wound healing models post-thaw [4].
Detailed Experimental Protocols
To ensure reproducible comparison across platforms, researchers should implement standardized evaluation protocols. The following methodologies are adapted from published studies comparing expansion systems and cryopreservation outcomes.
Protocol: Comparative Expansion of MSCs in Bioreactor vs TCP
Objective: To compare the expansion efficiency and resulting cell characteristics of MSCs grown in automated bioreactor systems versus traditional TCP flasks [4].
Materials:
- Starting Material: Bone marrow aspirate, adipose tissue, or umbilical cord tissue
- Culture Medium: MSC-qualified medium supplemented with human platelet lysate (preferred over FBS for GMP compliance) [39]
- Bioreactor System: Quantum, CliniMACS Prodigy, or Cocoon Platform with appropriate consumables
- Control System: TCP flasks (T175)
- Characterization Reagents: Flow cytometry antibodies for CD73, CD90, CD105, CD34, CD45, CD14; Differentiation kits for adipogenic, osteogenic, chondrogenic lineages
Methodology:
- Cell Isolation: Isolate MSCs from source tissue using collagenase digestion or density gradient centrifugation
- System Inoculation:
- For bioreactor systems: Follow manufacturer's instructions for seeding and system setup
- For TCP: Seed cells at 5,000 cells/cm² in T175 flasks
- Culture Conditions:
- Maintain at 37°C, 5% CO₂
- Use consistent medium composition across all systems
- For bioreactors: Follow automated medium exchange protocols
- For TCP: Perform medium changes every 2-3 days
- Harvesting:
- For bioreactors: Follow automated harvest procedures
- For TCP: Use trypsin/EDTA detachment
- Analysis:
- Count cells and calculate population doublings
- Assess viability via trypan blue exclusion
- Perform immunophenotyping by flow cytometry
- Evaluate differentiation potential using standard protocols
Protocol: Cryopreservation and Post-Thaw Functional Assessment
Objective: To evaluate the impact of cryopreservation on MSC characteristics expanded in different systems [4].
Materials:
- Cryopreservation Medium: Culture medium supplemented with 10% DMSO
- Controlled-Rate Freezer or freezing container
- Liquid nitrogen storage system
- Water bath (37°C for thawing)
- Wound healing assay reagents: Fibroblasts, migration assay materials
Methodology:
- Cryopreservation:
- Resuspend harvested MSCs in cryopreservation medium at 1-5 × 10⁶ cells/mL
- Transfer to cryovials and freeze using controlled-rate freezer (-1°C/min to -80°C, then transfer to liquid nitrogen)
- Store in liquid nitrogen vapor phase for minimum 1 week
- Thawing and Recovery:
- Rapidly thaw cryovials in 37°C water bath
- Transfer cells to pre-warmed culture medium
- Centrifuge and resuspend in fresh medium
- Plate at 5,000 cells/cm² and culture for 24 hours
- Post-Thaw Assessment:
- Viability: Measure using flow cytometry with viability dyes
- Immunophenotype: Analyze surface marker expression by flow cytometry
- Functionality:
- Trilineage differentiation: Culture in induction media for 2-3 weeks, stain with lineage-specific dyes
- Clonogenicity: Perform colony-forming unit (CFU) assay by plating at low density and counting colonies after 10-14 days
- Proliferation potential: Monitor growth kinetics over 7-10 days
- Immunomodulatory function: Measure T-cell suppression in co-culture assays
- Paracrine activity: Assess effects on fibroblast migration using scratch assay
Diagram 1: Experimental workflow for comparing TCP versus bioreactor-expanded MSCs after cryopreservation
Research Reagent Solutions
Successful implementation of comparative studies between automated platforms and traditional methods requires specific reagent systems optimized for each platform and research objective.
Table 3: Essential Research Reagents for Comparative MSC Studies
| Reagent Category | Specific Products | Function & Importance | GMP Compliance |
|---|---|---|---|
| Culture Medium | MSC-Brew GMP (Miltenyi), StemMacs MSC Expansion Media | Defined, xeno-free media supports regulatory compliance and consistency | GMP-grade available |
| Growth Supplement | Human Platelet Lysate (hPL) | Superior to FBS for MSC expansion; reduces immunogenicity concerns [39] | GMP-grade available |
| Bioreactor Coating | Recombinant fibronectin, vitronectin, cryoprecipitate | Essential for cell adhesion in hollow fiber bioreactors like Quantum [39] | GMP-grade available |
| Dissociation Reagents | Trypsin/EDTA, TrypLE Select, enzyme-free alternatives | Cell harvesting while maintaining viability and surface markers | GMP-grade available |
| Cryopreservation Medium | CryoStor CS10, STEM-CELLBANKER | Serum-free, defined formulations improve post-thaw recovery | GMP-grade available |
| Characterization Antibodies | CD73, CD90, CD105, CD34, CD45, CD14 (ISCT panel) | Essential for MSC identity verification and quality control [39] [4] | GMP-grade available |
| Differentiation Kits | Adipogenic, osteogenic, chondrogenic induction media | Functional validation of MSC multipotency [4] | Research-grade |
Platform Selection Framework
Choosing among these platforms depends on research objectives, scale requirements, and operational constraints. The following decision framework aids in platform selection:
Quantum System: Best suited for large-scale allogeneic MSC production where high cell yields are paramount. Its hollow fiber technology provides efficient expansion in a closed system, with demonstrated maintenance of MSC immunomodulatory function [39].
CliniMACS Prodigy: Ideal for laboratories requiring full automation from cell isolation through harvest, particularly when processing multiple cell types beyond MSCs. Its integrated design minimizes manual intervention [39].
Cocoon Platform: Optimal for decentralized manufacturing models and organizations requiring high protocol flexibility across different cell therapy products. Its customizable nature supports process development and clinical-scale production [42] [44].
Diagram 2: Decision framework for selecting an automated closed-system platform
The Quantum, CliniMACS Prodigy, and Cocoon platforms each offer distinct advantages for MSC manufacturing within the context of TCP versus bioreactor expansion research. Critical evaluation of experimental data reveals that while these automated systems significantly improve manufacturing efficiency and scalability compared to traditional TCP methods, they can influence MSC characteristics differently—particularly following cryopreservation. The Quantum system demonstrates superior yields for large-scale MSC production, the CliniMACS Prodigy offers complete automation from isolation to harvest, and the Cocoon Platform provides exceptional flexibility for customized processes. Research indicates that both TCP and bioreactor-expanded MSCs retain functionality after cryopreservation, albeit with potentially significant differences in immunophenotypic profiles that may influence therapeutic applications [4]. Researchers should select platforms based on specific research goals, regulatory requirements, and intended clinical applications, while continuing to monitor emerging data on how expansion methodologies influence the critical quality attributes of therapeutic MSCs.
The field of cell culture is undergoing a fundamental transformation, moving away from traditional, ill-defined supplements toward more precise and ethically conscious formulations. For decades, fetal bovine serum (FBS) has been the cornerstone supplement in culture media, providing a complex mixture of growth factors, hormones, and adhesion factors necessary for cell survival and proliferation. However, its undefined nature, significant batch-to-batch variability, and substantial ethical concerns related to animal welfare present major limitations for both research reproducibility and clinical applications [45] [46] [47]. These challenges, coupled with the stringent requirements of regulatory agencies for clinical-grade cell therapies, have accelerated the shift toward xeno-free and chemically defined media (CDM) [48] [47].
This transition is particularly critical for the manufacturing of mesenchymal stem/stromal cells (MSCs), which are promising candidates for regenerative medicine and cell-based therapies. The culture medium environment is a decisive factor that influences MSC characteristics, including their proliferation capacity, differentiation potential, immunomodulatory functions, and overall therapeutic efficacy [49] [50]. This guide provides a comparative analysis of FBS and its modern alternatives, presenting experimental data to inform researchers and developers in selecting the optimal culture system for their specific applications.
Classification and Definitions: Navigating the Terminology
Understanding the precise terminology is essential for selecting appropriate culture media. The following table clarifies the key definitions and compositions of different media types.
Table 1: Classification and Definitions of Cell Culture Media
| Media Type | Definition | Key Components | Regulatory & Scientific Advantages |
|---|---|---|---|
| Serum-Containing Media | Basal medium supplemented with animal serum (e.g., FBS). | Undefined mixture of growth factors, hormones, proteins, and other serum components [45]. | Established, broad compatibility; low cost. |
| Xeno-Free Media | Free of any components of non-human animal origin. | Human-derived supplements (e.g., Human Platelet Lysate) or recombinant factors [48] [50]. | Eliminates xenogeneic immune reactions; addresses ethical concerns. |
| Serum-Free Media (SFM) | Formulated without serum, but may contain purified proteins or animal-derived components. | Purified hormones, growth factors, and carrier proteins like albumin, which may be animal-sourced [45]. | Reduces batch variability compared to serum; more controlled than FBS. |
| Chemically Defined Media (CDM) | All components are known, identified chemicals of precise concentration; no biologicals of undefined composition. | Exact quantities of amino acids, vitamins, lipids, recombinant proteins, and other synthetic components [48] [49]. | Maximum batch-to-batch consistency; compliant with regulatory standards; eliminates risk of adventitious agents. |
A critical point of confusion in the field is the distinction between "Serum-Free" and "Chemically Defined." Research has revealed that some commercially available serum-free media (SFM) can still contain significant levels of human blood-derived components, such as myeloperoxidase, glycocalicin, and fibrinogen, essentially reclassifying them as human platelet lysate (hPL) from a compositional standpoint [45]. Therefore, researchers must scrutinize manufacturer claims and certificates of analysis to ensure media meet their specific requirements.
Comparative Performance Analysis of Media Supplements
Cell Growth and Proliferation
The performance of a culture medium is most fundamentally assessed by its ability to support robust cell expansion. Comparative studies consistently show that modern xeno-free and defined formulations can match or surpass the performance of traditional FBS.
Table 2: Comparative Performance of Media Supplements in MSC Culture
| Media Supplement | Reported MSC Expansion (vs. FBS control) | Key Growth Factors | Reported Impacts on MSC Function |
|---|---|---|---|
| Fetal Bovine Serum (FBS) | Baseline | Complex, undefined mixture | Standard reference, but associated with functional heterogeneity [49]. |
| Human Platelet Lysate (hPL) | Supports MSC growth effectively; often superior to FBS [45]. | PDGF, TGF-β1, VEGF, IGF-1 [45]. | Can introduce donor-dependent cytokines that skew immunomodulatory profiles [47]. |
| Xeno-Free/SFM (PRIME-XV) | Significantly faster proliferation up to 60 days in culture [50]. | Defined, proprietary composition. | Enhanced adipogenic differentiation and angiogenic activity [50]. |
| Chemically Defined Media (CDM) | Up to 4X higher growth over 3 passages [49]. | FGF-2, TGF-β1, EGF, PDGF-BB, IGF-1 [49]. | Enables standardized manufacturing; immunomodulatory function can be maintained or tuned [49]. |
A landmark high-throughput screening study identified specific CDM formulations that supported a four-fold increase in MSC growth over three passages compared to serum-containing media [49]. Similarly, adipose-derived stem cells (ASCs) cultured in a commercial xeno-free, serum-free medium (PRIME-XV XV) demonstrated significantly faster proliferation rates compared to those grown in FBS or hPL-supplemented media over a 60-day culture period [50].
Functional Properties and Therapeutic Potential
Beyond proliferation, the medium formulation profoundly impacts MSC functionality. A study on adipose-derived stem cells revealed that culture in xeno-free medium (XV) resulted in the most pronounced adipogenic differentiation and the highest angiogenic activity, as measured by endothelial cell network formation. This was associated with a 90-fold increase in hepatocyte growth factor (HGF) gene expression compared to FBS-cultured cells [50]. This suggests that medium selection can be tailored to specific clinical applications, such as reconstructive surgery.
Furthermore, CDM formulations have been shown to reduce variability in MSC bioactivity. By using a defined composition, the functional heterogeneity introduced by undefined supplements like FBS or hPL can be significantly minimized, leading to more consistent and predictable cell products [49].
Economic and Regulatory Considerations
From a cost perspective, human platelet lysate (hPL) currently offers a favorable cost-performance balance for research and clinical-scale expansions, being significantly more affordable than many SFM and CDM [45]. However, this must be weighed against its own batch variability challenges. While SFM and CDM are generally more expensive, their defined nature supports regulatory compliance by eliminating animal-derived components and reducing contamination risks, which is a critical requirement for clinical translation [48] [47]. The initial higher investment in defined media can be offset by reduced screening costs, improved process consistency, and lower failure rates in manufacturing.
Experimental Protocols for Media Evaluation
To objectively compare different media formulations, researchers can employ the following standardized experimental approaches.
Protocol for Growth Kinetics Analysis
Objective: To quantify the proliferation rate and maximum cell yield of MSCs in different media supplements.
- Cell Seeding: Thaw and pre-culture a standardized MSC batch (e.g., from adipose tissue or bone marrow). Seed cells at a defined density (e.g., 3,000 - 5,000 cells/cm²) in T-flasks or multi-well plates.
- Media Application: Apply the test media (e.g., FBS-supplemented, hPL-supplemented, CDM). Include appropriate coating (e.g., fibronectin for CDM) if required [49].
- Culture Maintenance: Culture cells over multiple passages (e.g., 3-5 passages). Perform medium exchanges according to manufacturer instructions or standard protocols (e.g., 80% exchange every 2-3 days) [51].
- Data Collection: At each passage, detach cells and perform viable cell counts using a trypan blue exclusion assay or an automated cell counter. Calculate population doublings (PD) and cumulative population doublings.
- Analysis: Plot growth curves and compare doubling time, maximal cell concentration, and total cell yield across the different media conditions [45] [49].
Protocol for Functional Characterization
Objective: To assess the impact of media on MSC differentiation potential and secretome.
- In Vitro Differentiation:
- Adipogenesis: Culture confluent MSCs in adipogenic induction medium for 14-21 days. Fix cells and stain with Oil Red O to visualize lipid droplets [4] [50].
- Osteogenesis: Culture confluent MSCs in osteogenic induction medium for 21-28 days. Fix cells and stain with Alizarin Red S to detect calcium deposits [4].
- Immunophenotyping:
- Secretome Analysis:
- Collect conditioned medium from MSC cultures.
- Analyze the concentration of key therapeutic factors (e.g., HGF, VEGF, IGF-1) using enzyme-linked immunosorbent assays (ELISA) [45] [50].
- Evaluate functional impact using an in vitro angiogenesis assay with Human Umbilical Vein Endothelial Cells (HUVECs) to assess capillary network formation [50].
The Scientist's Toolkit: Essential Reagents for Defined Culture
Transitioning to xeno-free and defined systems requires a suite of specific reagents.
Table 3: Essential Research Reagent Solutions for Xeno-Free and Defined Culture
| Reagent Category | Specific Examples | Function & Rationale | Animal-Free Alternative |
|---|---|---|---|
| Basal Media | DMEM/F12, α-MEM | Provides essential nutrients, vitamins, and salts. | Same (inherently chemical). |
| Growth Factors | Recombinant FGF-2, EGF, PDGF-BB, TGF-β1 [49]. | Stimulates proliferation and maintains cell functions. | Recombinant proteins (xeno-free). |
| Attachment Factors | Human Fibronectin, Recombinant Laminin [49]. | Coats surface to enable cell adhesion and spreading. | Human-sourced or recombinant proteins. |
| Detachment Agents | Recombinant TrypLE [46]. | Enzymatically dissociates adherent cells for passaging. | Recombinant fungal-origin enzymes. |
| Carrier Proteins | Recombinant Human Albumin, Synthetic Polymers. | Stabilizes other components, replaces animal-derived BSA. | Recombinant or synthetic. |
| Lipids & Micronutrients | Chemically Defined Lipid Concentrates, Soybean-derived Lipids. | Supplies cholesterol and fatty acids for membrane synthesis. | Plant-derived or synthetic. |
Visualizing the High-Throughput Screening Workflow
Advanced screening approaches are crucial for developing optimized CDM. The following diagram illustrates a high-throughput workflow that uses morphological profiling to identify media formulations that enhance both MSC growth and function.
Diagram 1: High-Throughput Media Screening Workflow.
This workflow enables the systematic and efficient screening of hundreds of growth factor combinations to identify optimal CDM formulations that support robust MSC expansion while maintaining critical therapeutic functions [49].
The collective evidence firmly establishes that xeno-free and chemically defined media are viable and superior alternatives to FBS for the manufacturing of MSCs. While FBS remains a common reagent in research, its undefined nature and ethical drawbacks make it unsuitable for clinical-grade cell production. The shift toward CDM represents the future standard for regenerative medicine, offering unparalleled consistency, regulatory compliance, and the ability to tailor media to enhance specific MSC functions.
Future developments will be driven by omics-based profiling (transcriptomics, proteomics, metabolomics) and artificial intelligence that can decode the complex nutritional requirements of cells and design optimized media formulations [47]. Furthermore, the creation of universal, human plasma-like media that more accurately mimics the human physiological environment will enhance the translational relevance of in vitro cultured cells [47]. As these innovations mature, they will further solidify the role of defined, ethical, and high-performance culture systems in advancing both basic research and clinical applications in cell therapy.
Navigating Challenges: Cryopreservation, Potency Loss, and Process Optimization
For researchers in cell therapy and regenerative medicine, the choice of cryopreservation protocol is a critical determinant of clinical success. The transition from laboratory research to clinical application hinges on the ability to preserve cellular integrity and functionality after thawing. Among the various cryopreservation methodologies, slow freezing and vitrification have emerged as the two predominant approaches, each with distinct physical mechanisms and biological consequences. Slow freezing involves controlled, gradual cooling that allows for extracellular ice formation and cellular dehydration, while vitrification utilizes high cooling rates and high cryoprotectant concentrations to achieve a glass-like solid state without ice crystal formation [52]. This comprehensive analysis examines the comparative effectiveness of these techniques across diverse cell types, with particular emphasis on their impact on mesenchymal stromal cells (MSCs) expanded in traditional tissue culture polystyrene (TCP) versus bioreactor systems—a critical consideration for scaling up clinical-grade cell manufacturing.
Fundamental Principles and Technical Mechanisms
Physicochemical Basis of Cryopreservation
The fundamental challenge in cryopreservation lies in navigating the physical transitions of water as temperatures decrease. During slow freezing, cells are subjected to a programmed cooling rate (typically -1°C/min to -5°C/min) in the presence of permeating cryoprotectants like dimethyl sulfoxide (DMSO) or ethylene glycol. This gradual cooling allows for controlled dehydration as ice forms primarily in the extracellular space, minimizing lethal intracellular ice formation. The process requires specialized, expensive programmable freezers but has been the conventional method for decades [53] [52].
In contrast, vitrification achieves ultra-rapid cooling rates (up to 20,000°C/min) using high concentrations of cryoprotectants (often 6-8 M), resulting in a dramatic increase in viscosity that prevents ice crystal formation entirely. Instead, the solution solidifies into a glass-like amorphous state through molecular immobilization. This method is notably faster and requires less expensive equipment, but introduces potential cytotoxicity concerns from high cryoprotectant concentrations and requires precise technical execution [53] [52].
Comparative Workflow Diagrams
The following workflow diagrams illustrate the fundamental procedural differences between these two cryopreservation approaches:
Figure 1: Comparative workflows for slow freezing versus vitrification protocols
Comparative Performance Analysis Across Cell and Tissue Types
Ovarian Tissue Cryopreservation
The cryopreservation of ovarian tissue presents unique challenges due to its complex multicellular architecture and density. A meta-analysis of 14 comparative studies revealed nuanced differences between the two methods. While the proportion of intact primordial follicles showed no significant difference between vitrification and slow freezing (OR = 0.98; 95% CI, 0.74-1.28), vitrification demonstrated significantly less DNA fragmentation in primordial follicles (RR = 0.71; 95% CI, 0.62-0.80; P < 0.00001) and better preservation of stromal cells (RR = 1.69; 95% CI, 1.47-1.94; P < 0.00001) [52].
Recent transplantation studies in nude mice further support these findings. After heterotopic transplantation of human ovarian tissue, the VF2 vitrification group showed significantly higher estradiol levels at 6 weeks post-transplantation compared to both the VF1 vitrification group and the slow freezing group (P < 0.05). Additionally, at 6 weeks post-transplantation, the proportion of normal follicles was significantly higher in the VF2 vitrification group compared to the slow freezing group (P < 0.05) [53].
Embryo and Gamete Cryopreservation
A systematic review and meta-analysis of cryopreserved human embryos demonstrated the superiority of vitrification for both cleavage-stage embryos and blastocysts. The survival rate of cleavage stage embryos was significantly higher after vitrification compared with slow freezing (odds ratio 15.57, 95% confidence interval 3.68-65.82). For blastocysts, vitrification also yielded significantly higher post-thaw survival rates (odds ratio 2.20, 95% confidence interval 1.53-3.16) [54].
Stem Cell and Hematopoietic Cell Cryopreservation
The impact of cryopreservation method varies significantly across different stem cell types. For hematopoietic stem cells (HSCs), a retrospective analysis of 72 cryopreserved products revealed that long-term storage at -80°C (uncontrolled-rate freezing) maintained median post-thaw viability at 94.8%, despite a moderate time-dependent decline of approximately 1.02% per 100 days (R2 = 0.283, p < 0.001) [55]. Engraftment kinetics were preserved in most patients, demonstrating that even suboptimal cryopreservation conditions can maintain sufficient HSC viability for clinical efficacy.
For adipose-derived stem cells (ASCs), the expansion system significantly influences cryopreservation outcomes. When ASCs were expanded in TCP versus hollow fiber bioreactor (HFB) systems and subsequently cryopreserved, notable differences emerged in immunophenotypic profiles. While CD73 and CD90 expression remained highly expressed (>95%) in both systems before and after freezing, CD105 expression in TCP-expanded cells significantly decreased from >95% to 75% after freeze-thawing, creating a significant difference between systems post-preservation [4].
Testicular Tissue Cryopreservation
A comparative analysis of neonatal bovine testicular tissue revealed method-specific structural preservation patterns. Vitrification resulted in a significantly lower proportion of seminiferous tubules with >70% attachment to the basement membrane (19.15 ± 1.82%) compared to both controlled slow freezing (47.89 ± 10.98%) and uncontrolled slow freezing (39.05 ± 4.15%) (P < 0.05). However, no significant differences were observed in the proportion of seminiferous tubules containing PGP9.5-positive germ cells across methods, and germ cell densities per unit area were comparable [56].
Table 1: Comparative Performance of Cryopreservation Methods Across Cell and Tissue Types
| Cell/Tissue Type | Performance Metric | Slow Freezing | Vitrification | Statistical Significance |
|---|---|---|---|---|
| Ovarian Tissue | DNA fragmentation in primordial follicles | Reference | RR = 0.71 (0.62-0.80) | P < 0.00001 [52] |
| Normal stromal cells | Reference | RR = 1.69 (1.47-1.94) | P < 0.00001 [52] | |
| Estradiol levels post-transplantation | Reference | Significantly higher | P < 0.05 [53] | |
| Human Embryos | Cleavage-stage survival rate | Reference | OR = 15.57 (3.68-65.82) | Significant [54] |
| Blastocyst survival rate | Reference | OR = 2.20 (1.53-3.16) | Significant [54] | |
| Testicular Tissue | Tubules with >70% basement membrane attachment | 39.05-47.89% | 19.15% | P < 0.05 [56] |
| HSCs | Post-thaw viability after long-term -80°C storage | 94.8% (median) | Not reported | Time-dependent decline [55] |
| ASCs (TCP-expanded) | CD105+ population pre-/post-cryopreservation | >95% / 75% | Similar pattern | Significant decrease [4] |
Impact of Cell Expansion Systems on Cryopreservation Outcomes
TCP vs. Bioreactor Expansion Systems
The expansion methodology employed prior to cryopreservation significantly influences post-thaw cell characteristics. A comparative analysis of adipose-derived stem cells (ASCs) expanded in tissue culture polystyrene (TCP) flasks versus hollow fiber bioreactor (HFB) systems revealed that cryopreservation differentially affects immunophenotypic subpopulations depending on the expansion platform [4].
Before cryopreservation, both TCP and HFB systems showed >95% expression of CD73, CD90, and CD105. However, after freeze-thawing, TCP-expanded cells exhibited a significant reduction in CD105 expression to 75%, while HFB-expanded cells maintained stable CD105 expression. This suggests that HFB expansion may confer greater resilience to the cryopreservation process for certain surface markers [4].
The two expansion systems also supported different immunophenotypic subpopulations, influencing heterogeneity within ASC cultures. Following cryopreservation, TCP-expanded cells became less variable while HFB-expanded cells became more variable during the freeze-thaw process. Despite these differences in subpopulation dynamics, both systems maintained comparable functional characteristics including trilineage differentiation capacity, clonogenicity, proliferation potential, and effects on fibroblast migration in wound healing assays [4].
Dynamic vs. Static Culture Systems
Three-dimensional culture systems introduce additional complexity to cryopreservation outcomes. Dynamic suspension cultures of MSC spheroids enable faster formation of more compact spheroids compared to static suspension cultures, while also achieving long-term maintenance of spheroid size and stemness properties [7]. The enhanced cell-cell and cell-extracellular matrix interactions in 3D cultures create a specialized niche that optimizes cellular function by mimicking the in vivo environment more closely than conventional 2D culture methods. These architectural differences likely influence cryoprotectant penetration and ice crystal formation during cryopreservation, though direct comparisons of cryopreservation outcomes between dynamic and static 3D cultures remain limited in the current literature.
Table 2: Impact of Expansion System on Post-Thaw ASC Characteristics [4]
| Parameter | TCP-Expanded ASCs | HFB-Expanded ASCs | Post-Thaw Change |
|---|---|---|---|
| CD73 Expression | >95% | >95% | Maintained in both |
| CD90 Expression | >95% | >95% | Maintained in both |
| CD105 Expression | >95% → 75% | >95% maintained | Significant decrease in TCP only |
| CD274 Expression | Higher pre-freeze | Lower pre-freeze | Balanced post-thaw |
| Subpopulation Heterogeneity | Decreased post-thaw | Increased post-thaw | Differential response |
| Viability | >90% | >90% | Comparable |
| Trilineage Differentiation | Maintained | Maintained | No functional difference |
| Clonogenicity | Maintained | Maintained (trend higher) | No significant difference |
| Fibroblast Migration Effect | Maintained | Maintained | Comparable functionality |
Experimental Protocols and Methodologies
Standardized Slow Freezing Protocol for Ovarian Tissue
The slow freezing method established by the American Medical Cooperative of Oncofertility Consortium involves placing ovarian tissue fragments in cryovials with a solution containing 1.5 M ethylene glycol and 1.0 M sucrose. A programmed freezing system is used with the following parameters: cooling at 2°C/min to -7°C, followed by a hold at this temperature for manual seeding, then continued cooling at 0.3°C/min to -40°C, and finally at 10°C/min to -140°C before transfer to liquid nitrogen for storage [57].
Vitrification Protocols for Ovarian Tissue
Two effective vitrification methodologies have been demonstrated for ovarian tissue cryopreservation:
Protocol VF1: Ovarian tissues are incubated in an equilibration solution containing 3.8% ethylene glycol, 0.5 M sucrose, and 6% synthetic serum substitute (SSS) for 3 minutes at room temperature, followed by transfer to 19% ethylene glycol and 0.5 M sucrose for 1 minute. Tissues are then incubated in vitrification solution (38% ethylene glycol, 0.5 M sucrose) for 11 minutes before plunging into liquid nitrogen [53].
Protocol VF2: This two-step protocol uses ascending concentrations of ethylene glycol and dimethylsulphoxide (DMSO). Ovarian tissues are placed in 10% ethylene glycol, 10% DMSO, and 20% SSS for 25 minutes at room temperature, followed by exposure to vitrification solution containing 20% ethylene glycol, 20% DMSO, 0.5 M sucrose, and 20% SSS for 15 minutes at room temperature before rapid cooling [53].
Post-Thaw Viability Assessment Methods
The accurate assessment of post-thaw viability requires method-specific considerations:
Acridine orange (AO) staining demonstrates greater sensitivity to delayed cellular degradation compared to 7-AAD flow cytometry, with a significant difference between methods (p < 0.001) observed in HSC viability assessments [55].
Flow cytometry with 7-AAD provides robust viability quantification but may underestimate delayed apoptosis compared to AO staining.
Functional assessments including clonogenicity assays, trilineage differentiation potential, and in vivo transplantation models provide critical validation beyond simple membrane integrity tests.
Decision Framework for Method Selection
The following decision pathway integrates multiple factors to guide researchers in selecting the appropriate cryopreservation method for their specific application:
Figure 2: Decision framework for selecting between slow freezing and vitrification methods based on cell type, resources, and desired outcomes
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Reagents and Materials for Cryopreservation Research
| Reagent/Material | Function | Example Application |
|---|---|---|
| Ethylene Glycol | Permeating cryoprotectant | Vitrification solutions (3.8-38%) [53] |
| Dimethyl Sulfoxide (DMSO) | Permeating cryoprotectant | Slow freezing (10%) and vitrification solutions [53] [56] |
| Sucrose | Non-permeating cryoprotectant | Osmotic buffer in both methods (0.1-1.0 M) [57] [53] |
| Synthetic Serum Substitute | Protein source | Prevents membrane damage during freezing (6-20%) [57] [53] |
| Acridine Orange/Propidium Iodide | Viability staining | Distinguishes live/dead cells post-thaw [55] |
| 7-AAD | Viability dye for flow cytometry | Membrane integrity assessment [55] |
| Programmable Freezer | Controlled-rate cooling | Slow freezing protocols [57] |
| Liquid Nitrogen Storage | Long-term preservation | Both methods (-196°C) [57] [53] |
| Cryovials | Sample containment | Both methods [57] [56] |
| Sterile Tissue Transport Medium | Pre-processing maintenance | HEPES-buffered M199 with antibiotics [53] |
The comparative analysis of slow freezing versus vitrification reveals a complex landscape where optimal method selection depends on specific cell types, analytical endpoints, and expansion systems. Vitrification generally demonstrates superior performance for embryos and gametes, with significantly higher survival rates, while its advantages for complex tissues include enhanced DNA integrity and stromal preservation. For MSC-based therapies, the expansion system significantly influences cryopreservation outcomes, with TCP-expanded cells showing greater susceptibility to surface marker alterations than bioreactor-expanded counterparts. As cell manufacturing evolves toward bioreactor-based production systems, understanding these interactions becomes increasingly critical for developing standardized, effective cryopreservation protocols that maintain therapeutic efficacy from benchtop to bedside.
Dimethyl sulfoxide (DMSO) has served as the cryoprotectant of choice for most animal cell systems since the early history of cryobiology, enabling stable and safe storage of bone marrow and blood cells for thousands of cell transplants that would not otherwise have been possible [58]. Despite its widespread use and exceptional effectiveness at preventing ice crystal formation, DMSO's effects on cell biology and apparent toxicity in patients have been an ongoing topic of debate for decades, driving the search for less cytotoxic alternatives [58]. This dilemma is particularly acute in the field of mesenchymal stem/stromal cell (MSC) therapies, where maintaining cell quality throughout the cryopreservation process is essential for therapeutic efficacy [32]. The fundamental challenge lies in balancing DMSO's proven protective capabilities against its documented adverse effects on cellular systems, a balance that requires careful consideration of concentration, exposure time, and cell-type specific vulnerabilities.
The toxicity profile of DMSO is dose-dependent, posing risks to both cellular function and patient safety [59]. On a cellular level, DMSO disrupts membrane integrity, interferes with mitochondrial function, and increases the production of reactive oxygen species (ROS), leading to oxidative damage [59]. Clinically, patients receiving cell therapy infusions containing DMSO have reported cardiovascular issues, neurological symptoms, gastrointestinal problems, allergic reactions, and hematological disturbances [59]. Contemporary research using sensitive high-throughput techniques has revealed that even relatively low concentrations of DMSO (0.1%) can induce large alterations in microRNAs and the epigenetic landscape, particularly in maturing cardiac models [60]. These findings challenge the traditional assumption that DMSO is biologically inert at commonly used concentrations.
Comparative Analysis of Cryoprotectant Options
Cryoprotectant Mechanisms and Toxicity Profiles
Cryoprotectants function through distinct mechanisms to protect cells during freezing and thawing cycles. Penetrating cryoprotectants like DMSO and glycerol enter cells and protect intracellular structures by lowering the freezing point and enabling vitrification, while non-penetrating agents like trehalose and sucrose primarily provide extracellular protection through water substitution, vitrification, and water entrapment mechanisms [59] [61].
Table 1: Comparison of Key Cryoprotectants in MSC Preservation
| Cryoprotectant | Mechanism of Action | Typical Concentration | Advantages | Disadvantages |
|---|---|---|---|---|
| DMSO | Penetrating; reduces ice crystal formation, stabilizes proteins | 5-10% (v/v) [59] | Highly effective, especially for mammalian cells; enables vitrification [59] | Dose-dependent cellular & patient toxicity; alters epigenome & transcriptome [59] [60] |
| Glycerol | Penetrating; lowers freezing point, stabilizes proteins & membranes | 5-15% (v/v) [59] | Lower toxicity than DMSO; ideal for RBCs & spermatozoa [59] | Can cause osmotic stress; requires tailored protocols for different cell types [59] |
| Trehalose | Non-penetrating; forms protective glass-like matrix, stabilizes biomolecules | 0.1-0.5 M [59] | Low toxicity, FDA GRAS status; consistent across temperatures [59] | Limited intracellular protection; often requires combination with penetrating agents [59] |
| Sucrose | Non-penetrating; osmotic buffer, membrane stabilizer | 0.1-0.5 M [59] | Low cytotoxicity, accessible, affordable [59] | Primarily extracellular protection; risk of osmotic shock with improper handling [59] |
| Bio-based Alternatives (AFPs, NADESs) | Varies by type: "adsorption-inhibition" (AFPs), colligative anti-freezing (NADESs) [61] | Varies | Biocompatible, low-toxicity, sustainable [61] | Emerging technology; not yet widely adopted in clinical settings [61] |
DMSO Toxicity: Molecular Insights and Functional Consequences
Recent research has revealed that DMSO induces drastic changes in human cellular processes even at low concentrations. Transcriptome analysis of 3D cardiac and hepatic microtissues exposed to 0.1% DMSO detected >2,000 differentially expressed genes affecting similar biological processes across tissue types, indicating consistent cross-organ actions of DMSO [60]. The most significantly affected pathways include cellular metabolism (particularly citric acid cycle and respiratory electron transport, glucose metabolism, and lipid metabolism) and vesicle-mediated transport (especially ER-to-Golgi anterograde transport and Golgi-mediated protein secretion) [60].
Perhaps more concerning are the effects of DMSO on the epigenetic landscape. Research demonstrates that DMSO exposure causes large-scale deregulations of cardiac microRNAs and significant alterations in genome-wide methylation patterns in cardiac microtissues, suggesting disruption of DNA methylation mechanisms [60]. These findings have particular implications for cryopreservation of embryos and oocytes, as DMSO may impact embryonic development through these epigenetic modifications.
Experimental Evidence: DMSO in MSC Cryopreservation
Impact of Expansion Systems on Post-Thaw MSC Characteristics
The choice between tissue culture polystyrene (TCP) and hollow fiber bioreactor (HFB) systems for MSC expansion has significant implications for how cells respond to cryopreservation with DMSO. A 2024 comparative analysis revealed that while both systems produced cells with >90% viability post-thaw, they exhibited important differences in surface marker expression after the freeze-thaw process [4].
Most notably, CD105 expression in TCP-expanded cells significantly decreased from >95% to 75% after freeze-thawing, whereas HFB-expanded cells maintained stable CD105 expression [4]. CD105 (endoglin) is a type I membrane glycoprotein essential for cell migration and angiogenesis, and its preservation is crucial for maintaining MSC therapeutic properties [10]. Additionally, the expression of CD274 (PD-L1, an immunomodulatory molecule) showed significant differences between the systems before freezing, but these differences balanced out post-thaw due to a nearly 48% increase in CD274 expression in HFB-expanded cells after cryopreservation [4].
The freeze-thaw process also drove differential changes in subpopulations between the expansion systems. Immunophenotypical analysis revealed that TCP-expanded cells became less variable and HFB-expanded cells more variable during cryopreservation, indicating that different culture conditions favor distinct subpopulations that respond differently to the stresses of freezing and thawing [4]. Despite these phenotypic changes, both expansion systems produced MSCs that retained their trilineage differentiation capacity and proliferation potential after thawing [4].
Table 2: Experimental Outcomes for TCP vs. HFB Expanded MSCs After Cryopreservation
| Parameter | TCP-Expanded MSCs | HFB-Expanded MSCs | Functional Significance |
|---|---|---|---|
| Viability | >90% post-thaw [4] | >90% post-thaw [4] | Meets minimal criteria for clinical applications |
| CD105 Expression | Significant decrease (from >95% to 75%) post-thaw [4] | Maintained stable expression post-thaw [4] | CD105 essential for cell migration & angiogenesis [10] |
| CD274 (PD-L1) Dynamics | Higher pre-freeze; stable post-thaw [4] | Lower pre-freeze; 48% increase post-thaw [4] | Immunomodulatory capacity may be affected |
| Population Heterogeneity | Became less variable after thawing [4] | Became more variable after thawing [4] | Different subpopulations may have varied therapeutic effects |
| Functional Characteristics | No significant difference in differentiation, proliferation, or wound healing effects [4] | No significant difference in differentiation, proliferation, or wound healing effects [4] | Core therapeutic potential maintained despite phenotypic changes |
Protocol Details: Cryopreservation Methods and Assessment
Standard cryopreservation protocols for MSCs typically involve slow freezing in the presence of 10% DMSO combined with autologous plasma or other protein sources, followed by storage at -80°C or in liquid nitrogen [13]. For example, in studies examining bone marrow aspirate concentrate (BMAC), the freezing medium consisted of 10% DMSO and 90% autologous plasma, with cooling in a controlled-rate container (~-1°C/min) before transfer to -80°C storage [13].
Advanced techniques to improve cryopreservation outcomes include the application of regulated shear stress in microfluidic bioreactors. Research demonstrates that with increase in shear stress, focal point adhesions (FPAs) increase between substrate and MSCs, and that increased FPAs (4e-3 μbar) enhance the cellular survivability of adherent MSCs after cryopreservation [6]. This approach represents a potentially new method for modifying cryopreservation protocols by up-regulating focal adhesion points of cells to improve clinical outcomes.
Assessment of post-thaw MSC function typically includes:
- Viability analysis via trypan blue exclusion or automated cell counting [4]
- Immunophenotyping for MSC markers (CD73, CD90, CD105) and hematopoietic markers (CD34, CD45, CD14, etc.) [4] [10]
- Functional assays including colony-forming unit fibroblasts (CFU-f) [13]
- Trilineage differentiation potential (adirogenic, osteogenic, chondrogenic) [4] [13]
- In vivo models for tissue repair capacity, such as OA rat models for cartilage repair [13]
Experimental Workflow: MSC Expansion to Post-Thaw Assessment
Emerging Solutions and Alternatives
DMSO-Free Cryopreservation Solutions
The need to balance DMSO efficacy with toxicity has driven the development of DMSO-free cryopreservation solutions. Recent advances include commercial serum-free, DMSO-free solutions that achieve similar cell recovery compared to DMSO-containing controls, as assessed by viability and viable cell density [32]. Importantly, MSCs cryopreserved with these alternative solutions show no loss in proliferative capacity compared to those preserved with DMSO-containing solutions when cultured post-thaw [32]. Nonclinical animal trials have demonstrated that these DMSO-free solutions are nontoxic, offering a viable alternative for cell therapy applications where DMSO toxicity is a concern [32].
Bio-based Cryoprotectants
Bio-based cryoprotectants represent another promising avenue for reducing DMSO dependence. These include antifreeze proteins (AFPs) and natural deep eutectic solvents (NADESs) that offer non-toxicity and biocompatibility [61]. AFPs from different sources change ice crystal morphology, exhibiting thermal hysteresis, and ice recrystallization inhibition (IRI) activity following an "adsorption-inhibition" mechanism [61]. NADESs show excellent capacities for freezing point depression and IRI due to their temperature-responded hydrogen-bonding supramolecular networks [61]. While these bio-based alternatives show great potential for future applications, they are not yet widely adopted in clinical MSC cryopreservation protocols.
Protocol Optimization Strategies
For researchers continuing to use DMSO, several strategies can help mitigate toxicity concerns:
- Minimize DMSO concentration and exposure time by using rapid cooling and warming protocols [59]
- Combine DMSO with non-penetrating cryoprotectants like trehalose or sucrose to reduce the required DMSO concentration while maintaining protection [59]
- Implement strict temperature control during addition and removal of DMSO, as its cytotoxicity increases with higher temperatures [59]
- Consider cell-specific sensitivities - human chondrocytes, oocytes, and certain immune cells are particularly sensitive to DMSO-induced toxicity and may require customized protocols [59]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for MSC Cryopreservation Research
| Reagent / Material | Function | Example Application |
|---|---|---|
| DMSO (High Purity) | Penetrating cryoprotectant | Standard cryopreservation at 5-10% concentration [59] |
| Trehalose | Non-penetrating cryoprotectant | Extracellular protection; combined with DMSO to reduce toxicity [59] |
| Serum-Free Cryopreservation Media | Defined formulation without animal components | Clinical-grade MSC preservation; reduces variability [62] [32] |
| Autologous Plasma | Protein source for freezing medium | BMAC cryopreservation (10% DMSO + 90% plasma) [13] |
| Controlled-Rate Freezer | Programmable cooling apparatus | Standardized freezing protocols (~-1°C/min) [6] [13] |
| Ficoll Gradient | Density separation medium | Mononuclear cell isolation from BMAC [13] |
| Flow Cytometry Antibodies | Cell surface marker detection | Immunophenotyping (CD73, CD90, CD105, CD34, CD45) [4] [10] |
| Trilineage Differentiation Kits | Differentiation potential assessment | Functional validation post-thaw [4] [13] |
Solution Strategies for the DMSO Dilemma
The cryoprotectant dilemma surrounding DMSO represents a classic challenge in translational science: balancing proven efficacy against documented toxicity. While DMSO remains the most effective cryoprotectant for many MSC applications, particularly in research settings, its significant effects on cellular processes—including alterations to the transcriptome, epigenome, and proteome—cannot be overlooked [60]. The differential impact of DMSO on cells from various expansion systems further complicates this picture, with TCP-expanded MSCs showing particular vulnerability to CD105 loss after freeze-thaw cycles [4].
For researchers and therapy developers, the path forward involves careful consideration of application-specific requirements. For critical clinical applications where functional properties must be perfectly preserved, emerging DMSO-free solutions offer promising alternatives [32]. For research applications where some functional alterations may be acceptable, DMSO remains a valuable tool when used with appropriate precautions. In all cases, comprehensive post-thaw characterization that goes beyond simple viability measures to include immunophenotyping, functional assays, and subpopulation analysis is essential for understanding the true impact of cryopreservation on MSC therapeutic potential.
The future of MSC cryopreservation will likely involve increasingly sophisticated combination approaches—utilizing reduced DMSO concentrations with complementary cryoprotectants, implementing physical optimization strategies like regulated shear stress, and eventually incorporating novel bio-based cryoprotectants as they transition from research curiosities to practical tools. Through these approaches, the field can maintain the benefits of DMSO while progressively mitigating its drawbacks, ultimately enhancing the safety and efficacy of MSC-based therapies.
Mitigating Expansion-Induced Senescence and Loss of Progenitor Potency
The transition of mesenchymal stem cells (MSCs) from laboratory research to clinical therapeutics necessitates large-scale expansion to achieve the millions to hundreds of millions of cells required per patient, depending on the disease and administration route [63]. However, in vitro expansion poses significant risks to cellular integrity, potentially inducing cellular senescence and diminishing the therapeutic potency that forms the basis for their clinical application [64]. This comparison guide objectively evaluates two primary expansion technologies—conventional tissue culture polystyrene (TCP) flasks and automated hollow fiber bioreactor (HFB) systems—with particular focus on their impact on cellular senescence and progenitor function following the critical process of cryopreservation.
The preservation of stem cell characteristics post-expansion and post-cryopreservation is paramount for clinical efficacy. Cellular senescence represents a state of stable cell cycle arrest triggered by various stressors, characterized by morphological changes, altered gene expression, and development of a pro-inflammatory senescence-associated secretory phenotype (SASP) [64] [65]. Senescent cells exhibit irreversible proliferative arrest while remaining metabolically active, secreting inflammatory cytokines, chemokines, growth factors, and proteases that can disrupt tissue microenvironment and compromise therapeutic potential [64]. This comprehensive analysis examines how different expansion systems either mitigate or exacerbate these detrimental processes, providing researchers with evidence-based guidance for manufacturing decisions.
Comparative Performance Analysis: TCP vs. Bioreactor-Based Expansion
Experimental Framework and Methodological Considerations
To ensure valid comparison between TCP and HFB expansion systems, researchers must establish equivalent experimental conditions despite fundamentally different passaging schedules. One validated protocol involves seeding one-fifth of adipose-derived stem cells (ASCs) in an HFB system (1.7 m²) for a single passage, while simultaneously seeding four-fifths of cells into T175 TCP flasks (0.175 m²) and expanding at a 1:3 ratio until passage 4, theoretically yielding 27 T175 flasks (totaling 0.47 m²) [4]. For practical considerations, one-third of TCP cells may be continued in each subsequent passage. HFB cells at P1 and TCP cells at P4 are then cryopreserved using standardized protocols for subsequent analysis [4].
Critical methodological considerations include:
- Population doubling equivalency: Ensure cells from both systems undergo similar population doublings before comparison
- Cryopreservation uniformity: Use identical cryoprotectant agents (typically 10% DMSO in autologous plasma or serum-containing medium) and controlled-rate freezing protocols
- Post-thaw assessment timing: Evaluate cells after equivalent post-thaw recovery periods (typically 24-48 hours)
- Donor-matched sources: Use cells from the same donor source when comparing expansion platforms
Table 1: Key Surface Marker Changes After Cryopreservation in Different Expansion Systems
| Surface Marker | TCP Pre-freeze | TCP Post-thaw | HFB Pre-freeze | HFB Post-thaw | Functional Significance |
|---|---|---|---|---|---|
| CD105 | >95% | ~75%* | >95% | >95% | Mesenchymal stem cell marker, angiogenesis |
| CD274 (PD-L1) | ~48% | ~48% | Significantly lower | Comparable to TCP | Immune modulation, checkpoint regulation |
| CD73, CD90 | >95% | >95% | >95% | >95% | Minimal impact from expansion or freezing |
| CD29, CD201 | ~100% | ~100% | ~100% | ~100% | Highly stable markers |
| CD34, CD45, CD31 | <9% | <9% | <9% | <9% | Hematopoietic/endothelial markers (negative) |
Note: Statistical significance (p < 0.05) for CD105 loss in TCP system post-thaw [4]
Immunophenotypic Stability and Subpopulation Dynamics
The immunophenotypic profile of MSCs, defined by the International Society for Cellular Therapy (ISCT) as ≥95% expression of CD73, CD90, and CD105 with ≤2% expression of hematopoietic markers (CD34, CD45, CD14, CD19, HLA-DR), represents a critical quality metric [10]. Comparative analyses reveal that while most surface markers remain stable through expansion and cryopreservation, significant differences emerge between expansion platforms in their resilience to freeze-thaw stress.
CD105 (endoglin), a type I membrane glycoprotein essential for cell migration and angiogenesis, demonstrates marked sensitivity to cryopreservation in TCP-expanded cells, dropping from >95% to approximately 75% positivity post-thaw [4]. In contrast, HFB-expanded cells maintain >95% CD105 expression through the freeze-thaw cycle, suggesting this platform better preserves structural and functional membrane integrity. CD274 (PD-L1), an immune checkpoint protein, shows significantly lower expression on HFB-expanded cells before freezing, but post-thawing, the proportion of CD274 positive cells becomes comparable between systems, indicating differential freezing-induced regulatory responses [4].
Subpopulation analysis reveals more profound system-specific differences. Two main subpopulations emerge based on CD73, CD90, and CD105 co-expression: SPA1 (CD73+CD90+CD105-) and SPA2 (CD73+CD90+CD105+). Before freezing, a significant difference exists between expansion systems for SPA2, which further enlarges post-thaw as SPA2 significantly decreases in TCP cells, directly corresponding to the loss of CD105 expression [4]. For HFB-expanded cells, ten meaningful subpopulations were identified based on CD166, CD200, CD34, CD146, CD271, CD274, and CD248 co-expression patterns, with TCP-expanded cells becoming less variable and HFB-expanded cells more variable during the freeze-thaw process [4].
Figure 1: Experimental workflow comparing TCP and HFB expansion systems with post-thaw assessment points. The diagram highlights key differences in how each system responds to the cryopreservation process, particularly regarding surface marker stability and population heterogeneity.
Functional Potency and Therapeutic Capacity
Beyond surface marker expression, the functional capacity of MSCs following expansion and cryopreservation represents the ultimate validation of manufacturing methodology. Critical functional assessments include trilineage differentiation potential (adiopogenic, osteogenic, chondrogenic), colony-forming unit (CFU) efficiency, proliferation kinetics, and paracrine signaling capacity through secreted factors.
Research demonstrates that both TCP and HFB-expanded MSCs maintain trilineage differentiation capability after cryopreservation, with positive staining for Oil Red O (adipocytes), Alizarin Red S (osteoblasts), and Alcian Blue (chondrocytes) [4]. Although HFB-cells appear to have higher colony-forming potential, this difference does not reach statistical significance. Similarly, proliferation kinetics show no significant difference between systems post-thaw, with both exhibiting characteristic growth curves and eventual detachment upon overconfluence due to crowding, waste accumulation, or nutrient depletion [4].
In wound healing assays that model therapeutic paracrine function, both manufacturing methods yield cells capable of influencing fibroblast migration and proliferation, key processes in tissue repair [4]. Importantly, cell viability exceeds 90% post-freeze-thaw in both systems, though TCP cells demonstrate greater robustness than HFB-cultured counterparts despite the more significant immunophenotypic shifts observed in TCP populations [4].
Table 2: Functional Characteristics of Cryopreserved MSCs from Different Expansion Systems
| Functional Parameter | TCP-Expanded | HFB-Expanded | Assessment Method | Clinical Significance |
|---|---|---|---|---|
| Trilineage Differentiation | Maintained | Maintained | Oil Red O, Alizarin Red S, Alcian Blue staining | Multipotency preservation |
| Colony-Forming Unit (CFU) Efficiency | Moderate | Higher (not statistically significant) | Crystal violet colony counting after 14 days | Stemness and self-renewal capacity |
| Proliferation Kinetics | No significant difference | No significant difference | Growth curve analysis over 11+ days | Expansion potential post-thaw |
| Post-Thaw Viability | >90% (more robust) | >90% | Trypan blue exclusion or flow cytometry | Clinical dosing accuracy |
| Paracrine Effects on Fibroblasts | Maintained | Maintained | Scratch/wound healing assay | Tissue repair capacity |
| Senescence Markers | System-dependent | System-dependent | SA-β-Gal, p16, p21, γH2AX | Therapeutic longevity |
Molecular Mechanisms of Expansion-Induced Senescence
Pathways to Senescence in Cultured MSCs
Cellular senescence in expanded MSCs can follow multiple molecular pathways, primarily driven by DNA damage response, oxidative stress, mitochondrial dysfunction, and epigenetic alterations [64] [66]. Replicative senescence occurs due to telomere erosion with successive population doublings, while stress-induced premature senescence can be triggered by suboptimal culture conditions regardless of division history [64].
Key molecular indicators of senescence include:
- Cell cycle inhibitors: Elevated p16INK4a, p21CIP1, p27, p19ARF, p53, and PAI-1
- DNA damage markers: Phosphorylated histone H2AX (γH2AX) at Ser139
- Senescence-associated β-galactosidase (SA-β-Gal): Increased lysosomal mass and activity
- Nuclear changes: Downregulation of lamin B1 protein, enlarged and flattened morphology
- SASP factors: Secretion of IL-6, IL-1, IL-8, TNF-α, and various proteases [64] [65]
The senescence-associated secretory phenotype (SASP) represents a particularly concerning aspect for therapeutic applications, as these secreted factors can create a pro-inflammatory microenvironment that may counteract the native immunomodulatory functions of MSCs and potentially promote pathological processes in recipient tissues [64] [65].
Bioreactor-Generated Mechanical Forces and Cellular Stress
HFB systems introduce dynamic fluid flow and shear stress that significantly influence MSC biology. Research indicates that regulated shear stress within specific parameters (0.002-0.004 μbar) enhances focal adhesion points (FPAs) between MSCs and substrate, with vinculin expression significantly higher under fluidic shear stress compared to static TCP controls [6]. This mechanical stimulation activates mechanotransduction pathways that influence cytoskeletal organization and cell signaling.
Interestingly, optimized shear stress appears to confer cryoprotective benefits, with increased FPAs correlating with enhanced post-thaw viability. This suggests that mechanical conditioning during expansion may strengthen structural integrity and stress resilience [6]. However, excessive shear stress can induce oxidative stress and premature senescence, highlighting the importance of parameter optimization in bioreactor systems.
Figure 2: Molecular pathways through which expansion culture conditions induce cellular senescence in MSCs. Multiple stressors converge on key mediator pathways that ultimately drive permanent cell cycle arrest and functional decline, compromising therapeutic utility.
Research Reagent Solutions for Senescence Mitigation
Table 3: Essential Research Tools for MSC Senescence and Potency Assessment
| Reagent/Category | Specific Examples | Research Application | Mechanistic Role |
|---|---|---|---|
| Senescence Biomarkers | SA-β-Gal staining, p16INK4a, p21, p53, γH2AX, Lamin B1 | Detection and quantification of senescent cells | Cell cycle arrest verification, DNA damage assessment |
| Surface Marker Antibodies | CD73, CD90, CD105, CD34, CD45, HLA-DR, CD274 | Immunophenotyping by flow cytometry | ISCT criteria verification, potency correlation |
| Cryoprotectants | DMSO, trehalose, autologous plasma, ectoine, catalase | Cryopreservation protocol optimization | Membrane stabilization, ice crystal inhibition, oxidative protection |
| Differentiation Kits | Adipogenic (Oil Red O), Osteogenic (Alizarin Red S), Chondrogenic (Alcian Blue) | Functional potency assessment | Multilineage capacity verification |
| SASP Analysis | IL-6, IL-1, IL-8, TNF-α, MMPs multiplex assays | Paracrine function evaluation | Inflammation modulation capacity |
| Cell Culture Media | αMEM with FBS/HPL, FGF-2 supplementation | Expansion condition optimization | Proliferation enhancement, senescence delay |
Strategic Implementation for Clinical Manufacturing
The selection between TCP and HFB expansion systems involves balancing multiple factors including scalability, reproducibility, phenotypic stability, and functional potency. TCP systems offer cost-effectiveness and versatility for smaller-scale research but present limitations in scalability and manual handling variability [4]. HFB systems excel in scalability and consistency for clinical manufacturing but require substantial initial investment and technical expertise [4] [63].
For clinical applications where CD105 expression is functionally important—particularly for therapies targeting angiogenic pathways—HFB expansion demonstrates clear advantages in preserving this critical marker through cryopreservation. Similarly, for applications requiring consistent cell populations, HFB systems provide more reproducible manufacturing outcomes. However, for research settings or applications where cost considerations predominate, TCP systems remain viable with awareness of their specific limitations in phenotypic stability.
Critical considerations for manufacturing strategy include:
- Therapeutic mechanism: Match expansion system to specific functional attributes required
- Scale requirements: Align manufacturing platform with cell quantity needs
- Regulatory pathway: Consider comparability data requirements for process changes
- Cryopreservation integration: Select systems that maintain critical quality attributes post-thaw
- Monitoring strategy: Implement comprehensive senescence and potency assessment
The integration of senescence mitigation strategies—including antioxidant supplementation, optimized shear stress, careful passage number limits, and validated cryopreservation protocols—ensures that manufactured MSCs maintain their functional potency from expansion through delivery to patients. As the field advances, continued refinement of these manufacturing platforms will further enhance the therapeutic reliability and clinical impact of MSC-based therapies.
The transition from cryopreserved cellular product to functionally active therapeutic represents one of the most vulnerable phases in mesenchymal stem cell (MSC) therapy. Post-thaw recovery protocols, particularly thawing methods and cryoprotectant agent (CPA) removal strategies, directly determine cell viability, functionality, and ultimately, therapeutic efficacy. As the field advances toward "off-the-shelf" cell therapies, standardized and optimized post-thaw processing has become indispensable for clinical and commercial success [67] [21]. This process is especially critical when considering MSCs expanded through different platforms—conventional tissue culture polystyrene (TCP) versus hollow fiber bioreactor (HFB) systems—as these expansion methods can impart distinct biological characteristics that may influence cryopreservation resilience [4].
The post-thaw phase encompasses two technically demanding procedures: the thawing process itself, which must rapidly transition cells from frozen to liquid state while minimizing ice recrystallization damage; and CPA removal, which must carefully manage osmotic stress during the dilution and removal of potentially toxic protective agents like dimethyl sulfoxide (DMSO) [21] [19]. Both procedures require precise execution to preserve the therapeutic properties of MSCs, including their immunomodulatory capacity, differentiation potential, and secretory functions [17] [68]. This guide systematically compares current methodologies, providing experimental data and protocols to inform research and development decisions for MSC-based therapies.
Thawing Methodologies: Technical Comparison
Fundamental Principles and Standardized Protocols
The thawing process aims to rapidly transition cells from -196°C (liquid nitrogen storage) to physiological temperatures, thereby minimizing the damaging effects of ice recrystallization during the phase change. The consistent application of rapid thawing in a 37°C water bath until only a small ice crystal remains is considered the gold standard across most cryopreservation studies [21]. This method typically achieves warming rates exceeding 100°C per minute, effectively surpassing the dangerous temperature range (-50°C to 0°C) where recrystallization occurs most readily [21].
Recent investigations have highlighted contamination risks associated with water bath thawing, prompting the development of alternative systems employing dry heat technology that maintain both rapid warming and closed-system sterility [21]. These systems are particularly valuable in Good Manufacturing Practice (GMP) environments where contamination control is paramount. Regardless of the heating method, the subsequent handling of cells after thawing—including CPA removal and resuspension—must be optimized to maintain cell viability and function, with research indicating that temperature maintenance at room temperature or 4°C during processing can significantly influence recovery metrics [19].
Comparative Analysis of Thawing Method Performance
Table 1: Comparison of Thawing Method Characteristics
| Thawing Method | Warming Rate | Contamination Risk | Scalability | Cell Viability Range | Best Applications |
|---|---|---|---|---|---|
| 37°C Water Bath | >100°C/min | High (without sealed container) | Moderate | 70-90% [21] [19] | Research labs, small-scale applications |
| Dry Thawing Equipment | ~50-80°C/min | Low | High | 75-90% [21] | GMP facilities, clinical applications |
| Direct Transfer to Media | <10°C/min | Low | Low | 50-70% [21] | Specialized protocols only |
CPA Removal Strategies: Technical Comparison
Fundamental Principles of CPA Removal
The removal of CPAs, particularly DMSO, presents a critical challenge in post-thaw processing. During this phase, cells face osmotic stress as extracellular osmolarity decreases rapidly, causing water influx that can lead to cell swelling and membrane rupture [21] [19]. The standard approach involves centrifugal washing to remove and dilute DMSO, but this process itself can cause significant cell loss due to the combined effects of osmotic stress and mechanical damage [21].
The toxicity of DMSO, while reduced at lower temperatures, remains a concern both for cell viability and potential patient side effects when administered clinically [69] [19]. Consequently, research has focused on developing gentler removal techniques and reducing initial DMSO concentrations through advanced cryopreservation strategies, including the use of biomaterials and intracellular-like cryoprotectant cocktails [69] [19].
Dilution-Based CPA Removal Strategies
Progressive dilution methods have emerged as a superior alternative to direct centrifugal washing, particularly for clinical applications where high cell concentrations are required. Research by Tan et al. demonstrated that cryopreserving MSCs at high concentrations (9 million cells/mL) followed by 1:2 dilution with Plasmalyte A/5% human albumin after thawing significantly improved viability maintenance over 6 hours compared to undiluted samples [19].
This approach allows for a more gradual osmotic adjustment, reducing the sudden volume changes that damage cell membranes. The study systematically compared different dilution strategies and found that cells cryopreserved at 6 million/mL with 1:1 dilution or 9 million/mL with 1:2 dilution (both achieving final concentration of 3 million/mL) showed better preservation of viability and recovery post-thaw [19]. The composition of the dilution medium also proves critical, with Plasmalyte A supplemented with 5% human albumin providing both osmotic stability and membrane protective effects [19].
Advanced CPA Removal Technologies
Microfluidic systems represent the technological frontier in CPA removal, offering precise control over the osmotic environment during DMSO dilution. The 3D micromixer technology developed by researchers enables continuous, automated mixing of cells with CPA solutions, achieving more homogeneous dilution than manual methods [70]. This system demonstrated excellent preservation of MSC viability, surface markers, differentiation potential, and proliferation capacity post-thaw, while eliminating human error and reducing contamination risk [70].
Another innovative approach involves hydrogel microencapsulation, where cells are encapsulated in alginate-based microspheres before cryopreservation. This technique provides a physical barrier that reduces osmotic stress during both freezing and thawing, allowing for a substantial reduction in DMSO concentration (as low as 2.5%) while maintaining viability above the 70% clinical threshold [69]. The microencapsulation technique not only mitigates cryoinjury but also maintains MSC phenotype and differentiation potential after thawing [69].
Comparative Analysis of CPA Removal Strategies
Table 2: Performance Comparison of CPA Removal Methods
| CPA Removal Method | Osmotic Stress | Cell Loss Percentage | DMSO Concentration After Removal | Processing Time | Suitable for Scale-Up |
|---|---|---|---|---|---|
| Direct Centrifugation | High | 15-30% [21] | <1% | Short | Moderate |
| Progressive Dilution | Moderate | 10-20% [19] | 3-5% (prior to infusion) | Medium | High |
| Microfluidic Systems | Low | 5-15% [70] | <0.5% | Short (continuous) | High |
| Microencapsulation | Very Low | <10% [69] | <2.5% (initial concentration) | Long (includes encapsulation) | Moderate |
TCP vs. HFB Expanded MSCs: Differential Post-Thaw Responses
Comparative Recovery and Functionality
The expansion system—TCP versus HFB—impacts MSC characteristics and their response to cryopreservation and post-thaw recovery. A comparative study examining ASCs from both systems revealed that despite generally maintained functionality after thawing, TCP-expanded cells demonstrated greater robustness in viability maintenance compared to HFB-cultured counterparts, with both systems exceeding 90% post-thaw survival [4].
The freeze-thaw process differentially affected certain surface markers between the systems. Specifically, CD105 expression significantly decreased in TCP-expanded cells after thawing, while CD274 expression patterns equilibrated between systems post-thaw despite initial differences [4]. These findings highlight the importance of considering expansion methodology when developing post-thaw protocols, as cells from different expansion platforms may require tailored approaches to optimize recovery of therapeutically relevant subpopulations.
Implications for Post-Thaw Processing
The observed differences between TCP and HFB-expanded MSCs suggest that standardized post-thaw protocols may need optimization based on the expansion platform. HFB systems, while offering advantages in scalability and consistency, may produce cells with different membrane composition or metabolic states that influence their response to osmotic stress during CPA removal [4]. This may necessitate adjustments in dilution rates, centrifugation forces, or recovery media composition to maximize functional cell recovery.
Experimental Protocols for Method Comparison
Standardized Thawing and Dilution Protocol
A rigorously tested protocol for thawing and CPA removal demonstrates the integration of multiple optimized steps [19]:
- Thawing: Remove vials from liquid nitrogen storage and immediately place in a 37°C water bath for approximately 2 minutes, gently agitating until only a small ice crystal remains.
- Surface Decontamination: Wipe vial exterior with 70% ethanol before transferring to biological safety cabinet.
- Controlled Dilution: For cells cryopreserved at high concentrations (6-9 million cells/mL), immediately add dropwise an equal volume (1:1) or double volume (1:2) of pre-warmed (room temperature) dilution medium (Plasmalyte A supplemented with 5% human albumin) with gentle mixing.
- Centrifugation: Centrifuge at 300-400 × g for 5 minutes at room temperature.
- Resuspension: Carefully decant supernatant and resuspend cell pellet in appropriate culture medium or infusion solution.
- Viability Assessment: Perform cell counting and viability assessment using Trypan blue exclusion or flow cytometry with Annexin V/PI staining.
This protocol has demonstrated maintenance of MSC surface marker expression (CD73, CD90, CD105), differentiation potential, and immunomodulatory functions post-thaw [19].
Microencapsulation Cryopreservation Protocol
For research exploring DMSO reduction strategies, the following microencapsulation protocol has proven effective [69]:
- Cell Preparation: Culture MSCs to 80% confluence, trypsinize, and collect cell pellet via centrifugation.
- Encapsulation Solution Preparation: On ice, prepare core solution containing 0.68g mannitol and 0.15g hydroxypropyl methylcellulose in sterile water.
- Cell Encapsulation: Resuspend MSC pellet in core solution and load into high-voltage electrostatic coaxial spraying device.
- Microsphere Generation: Use electrostatic spraying at 6kV with core solution flow rate of 25μL/min and sodium alginate shell solution at 75μL/min into calcium chloride solution for crosslinking.
- Cryopreservation: Transfer microspheres to cryopreservation solution containing 2.5% DMSO, then implement controlled-rate freezing to -80°C before liquid nitrogen storage.
- Post-Thaw Recovery: Thaw rapidly at 37°C, gently wash microspheres to remove CPA, and release cells via alginate dissolution or direct culture in microspheres.
This approach has enabled reduction of DMSO to 2.5% while maintaining viability above the 70% clinical threshold and preserving multilineage differentiation potential [69].
Visualizing Workflows and System Interactions
Post-Thaw Processing Workflow
Post-Thaw Processing Workflow
Osmotic Stress Management During CPA Removal
Osmotic Stress Management During CPA Removal
Essential Research Reagents and Solutions
Table 3: Essential Reagents for Post-Thaw Recovery Studies
| Reagent/Solution | Composition | Function | Example Application |
|---|---|---|---|
| Plasmalyte A/5% HA | Plasmalyte A, 5% Human Albumin | Dilution medium for osmotic stabilization | Progressive dilution of high-concentration cryopreserved cells [19] |
| NutriFreez D10 | Proprietary formulation with 10% DMSO | Commercial cryopreservation solution | Maintains viability and recovery post-thaw [19] |
| CryoStor CS5/CS10 | 5% or 10% DMSO in optimized base | Defined-composition cryopreservation solutions | Enables DMSO concentration comparison studies [19] |
| Sodium Alginate Solution | 0.2% sodium alginate, 0.46g mannitol | Hydrogel formation for microencapsulation | Enables low-DMSO cryopreservation [69] |
| Calcium Chloride Crosslinking | 6.0g calcium chloride in sterile water | Ionic crosslinking for hydrogel solidification | Microsphere formation in encapsulation protocols [69] |
| Annexin V/PI Staining | Annexin V-FITC, Propidium Iodide | Apoptosis and necrosis detection | Flow cytometry viability assessment post-thaw [19] |
Optimizing post-thaw recovery requires careful consideration of both thawing methodologies and CPA removal strategies, with selection dependent on specific research or clinical objectives. For most standard applications, rapid thawing at 37°C followed by progressive dilution centrifugation provides a robust balance of efficiency and effectiveness. For advanced applications aiming to minimize DMSO exposure or enhance scalability, microfluidic mixing and microencapsulation technologies offer promising alternatives with demonstrated efficacy in preserving MSC functionality.
The expansion system—TCP versus HFB—introduces another variable in post-thaw optimization, with evidence suggesting differential marker expression and potentially distinct responses to osmotic stress during CPA removal. Future protocol development should account for these biological differences to maximize recovery of therapeutically functional MSCs regardless of production platform. As cryopreservation science advances, continued refinement of these critical post-thaw processes will enhance the reliability, efficacy, and safety of MSC-based therapies.
Head-to-Head Validation: Phenotypic and Functional Potency of Post-Thaw MSCs
The therapeutic application of human mesenchymal stem cells (MSCs) necessitates in vitro expansion to achieve clinically relevant cell numbers, followed by cryopreservation to create "off-the-shelf" products that ensure immediate availability for treatments [63] [71]. The minimal criteria for defining MSCs, as established by the International Society for Cellular Therapy (ISCT), mandate a characteristic immunophenotype, specifically positive expression (≥95%) of the surface markers CD73, CD90, and CD105, in the absence of hematopoietic markers [10] [72]. Maintaining this phenotype after the freeze-thaw process is therefore a critical quality attribute for clinical efficacy and regulatory compliance.
The expansion methodology prior to cryopreservation is a significant variable. Traditional tissue culture polystyrene (TCP) flasks and automated bioreactor systems, such as the hollow fiber bioreactor (HFB), present distinct microenvironments that can influence cell characteristics [4] [73]. This guide provides a direct, data-driven comparison of the stability of the canonical MSC surface markers CD73, CD90, and CD105 following cryopreservation, specifically contrasting cells expanded in TCP versus bioreactor systems, to inform preclinical research and clinical manufacturing strategies.
Comparative Data Analysis: Post-Thaw Marker Stability
A direct comparative study investigated the immunophenotype of adipose-derived MSCs (ASCs) expanded in TCP and a Hollow Fiber Bioreactor (HFB) both before and after cryopreservation [4]. The findings indicate that the expansion system can significantly influence the stability of certain markers post-thaw.
Table 1: Impact of Expansion System and Cryopreservation on MSC Marker Expression
| Surface Marker | Expansion System | Pre-Freeze Expression (% Positive) | Post-Thaw Expression (% Positive) | Statistical Significance of Change |
|---|---|---|---|---|
| CD73 | TCP | > 95% | > 95% | Not Significant |
| HFB | > 95% | > 95% | Not Significant | |
| CD90 | TCP | > 95% | > 95% | Not Significant |
| HFB | > 95% | > 95% | Not Significant | |
| CD105 | TCP | > 95% | ~75% | Significantly Decreased |
| HFB | > 95% | > 95% | Not Significant |
The data reveals that while CD73 and CD90 expression remains highly stable (>95%) post-thaw regardless of the expansion system, CD105 stability is compromised specifically in TCP-expanded cells [4]. The expression of CD105 on TCP-expanded MSCs dropped significantly from over 95% to approximately 75% after the freeze-thaw procedure. In contrast, HFB-expanded cells maintained CD105 expression above 95% post-thaw. This suggests that the HFB system may confer a protective effect on the CD105 epitope or select for a subpopulation more resistant to cryopreservation-induced damage.
Further analysis of immunophenotypical subpopulations showed a significant increase in the CD73+/CD90+/CD105- subset and a corresponding decrease in the triple-positive (CD73+/CD90+/CD105+) population in TCP-expanded cells after thawing, confirming the specific loss of CD105 [4]. Despite these phenotypic shifts, the study confirmed that the fundamental stem cell characteristics, including trilineage differentiation potential and clonogenicity, were retained post-thaw in cells from both expansion systems [4].
Detailed Experimental Protocols from Key Studies
Protocol 1: Comparative Expansion and Cryopreservation of ASCs in TCP vs. HFB
This protocol is derived from the study that provided the core comparative data in Section 2 [4].
- 1. Cell Source and Baseline Culture: Human adipose-derived stem cells (ASCs) are isolated from lipoaspirate tissue and initially cultured in standard TCP flasks.
- 2. Scale-Up Expansion:
- TCP Group: Cells are seeded in T175 flasks and passaged at a 1:3 ratio upon confluence until passage 4 (P4). This schedule is designed to achieve a cumulative population doubling comparable to the HFB group.
- HFB Group: A single passage is conducted in a hollow fiber bioreactor system with a 1.7 m² surface area.
- 3. Cryopreservation: Expanded cells from both systems are harvested and cryopreserved using a DMSO-based cryoprotectant. The standard protocol involves cooling in a controlled-rate freezer to -90°C before transfer to the vapor phase of liquid nitrogen for storage.
- 4. Thawing and Analysis: Frozen vials are thawed in a 37°C water bath. Cells are then immediately processed for:
- Flow Cytometry: Cells are stained with antibodies against CD73, CD90, and CD105 (along with other markers) and analyzed on a flow cytometer. A minimum of 20,000 events are typically recorded.
- Functional Assays: Post-thaw cells are assessed for viability (e.g., using 7-AAD), trilineage differentiation (osteogenic, adipogenic, chondrogenic), and colony-forming unit (CFU) potential.
Protocol 2: Post-Thaw Reconstitution for Optimal Cell Recovery
This protocol focuses on a critical, often overlooked, step that directly impacts post-thaw viability and marker stability [74].
- 1. Thawing: Rapidly thaw cryopreserved MSC aliquots in a 37°C water bath.
- 2. Dilution and Washing: Immediately upon thawing, dilute the cell suspension drop-wise with a pre-warmed protein-containing solution. The study identified that using protein-free solutions (e.g., PBS alone) leads to significant cell loss (up to 50%).
- 3. Reconstitution: Centrifuge the diluted cells and resuspend the pellet in an appropriate vehicle for administration or further analysis. The study found that:
- Critical Parameter: Avoid reconstituting cells at concentrations below 10⁵ cells/mL in protein-free vehicles, as this causes instant cell loss.
- Optimal Solution: For post-thaw storage up to 4 hours, isotonic saline with 2% Human Serum Albumin (HSA) proved optimal, maintaining >90% viability with minimal cell loss.
- 4. Post-Thaw Storage: If immediate analysis or use is not possible, keep the reconstituted cells at room temperature and process within the validated stability window (e.g., 4 hours for saline with HSA).
Visualizing Experimental Workflows and Signaling Pathways
Experimental Workflow for Comparative Immunophenotyping
The following diagram outlines the logical flow of a standard experiment designed to compare post-thaw marker stability between TCP and bioreactor-expanded MSCs.
Signaling Pathways of Characteristic Surface Markers
The canonical MSC markers are not merely identifiers; they are functional proteins involved in key signaling pathways that govern MSC activity. The diagram below illustrates their primary roles.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for MSC Expansion, Cryopreservation, and Characterization
| Reagent / Material | Function in Experimental Context | Key Consideration |
|---|---|---|
| Hollow Fiber Bioreactor (HFB) | Provides a large surface-to-volume ratio for scalable 3D cell expansion in a controlled environment. | Mimics in vivo conditions more closely than TCP; requires significant initial investment and technical expertise [4] [63]. |
| Tissue Culture Polystyrene (TCP) Flasks | Standard 2D substrate for MSC expansion. Cost-effective and versatile for small-scale research. | Labor-intensive at large scale; may induce senescence and phenotypic instability at higher passages [4] [73]. |
| Dimethyl Sulfoxide (DMSO) | Penetrating cryoprotectant agent (CPA) that prevents intracellular ice crystal formation during freezing. | Cytotoxic at room temperature; must be removed post-thaw. Clinical-grade, low-endotoxin versions are required for therapeutics [75] [74]. |
| Human Serum Albumin (HSA) | Protein additive used in thawing and reconstitution solutions. Prevents cell loss and maintains viability post-thaw. | Critical for mitigating the "dilution shock" when reconstituting cryopreserved cells; use clinical-grade for translational work [74]. |
| Flow Cytometry Antibodies (CD73, CD90, CD105) | Fluorescently-labeled antibodies for quantifying surface marker expression via flow cytometry. | Conjugation to different fluorochromes allows for simultaneous multi-parameter analysis. Must be titrated for optimal signal-to-noise ratio [75] [4]. |
| Human Platelet Lysate (hPL) | Serum-free, xeno-free supplement for MSC culture media. Promotes robust proliferation. | Can replace fetal bovine serum (FBS), reducing immunogenic risk and variability for clinical manufacturing [63]. |
The stability of the definitive MSC immunophenotype, defined by CD73, CD90, and CD105, after cryopreservation is not guaranteed and is significantly influenced by the pre-freeze expansion platform. The consensus from comparative data indicates that CD73 and CD90 are highly robust markers, largely unaffected by the expansion system or the freeze-thaw process. In contrast, CD105 demonstrates marked vulnerability to cryopreservation-induced loss, but specifically in MSCs expanded in traditional TCP systems.
This evidence strongly suggests that bioreactor-based expansion systems, such as the hollow fiber bioreactor, may provide a more favorable microenvironment for maintaining the genomic and phenotypic stability of MSCs through the rigors of scale-up and cryopreservation. For researchers and clinicians, this necessitates a critical evaluation of the expansion methodology when designing production processes for cryopreserved MSC-based therapies. Ensuring that the final cell product not only meets but stably retains the ISCT criteria post-thaw is paramount for predicting in vivo efficacy and ensuring patient safety. Future work should focus on elucidating the molecular mechanisms behind CD105's instability and further optimizing bioreactor conditions and cryopreservation protocols to enhance the resilience of this critical marker.
In the field of regenerative medicine, mesenchymal stem cells (MSCs) represent a cornerstone for cellular therapy and tissue engineering applications. A critical challenge in their clinical translation lies in the requirement for large-scale expansion and subsequent cryopreservation, processes that can significantly impact their fundamental biological properties. This guide provides a systematic comparison of two primary expansion methodologies—tissue culture polystyrene (TCP) and hollow fiber bioreactor (HFB) systems—with a specific focus on their functional potency post-cryopreservation, as measured through trilineage differentiation potential and colony-forming efficiency. The preservation of these properties following freeze-thaw cycles is paramount for developing effective "off-the-shelf" cellular therapeutics, yet the influence of different expansion platforms remains incompletely characterized. By synthesizing current experimental data and standardizing assessment protocols, this analysis aims to support researchers and drug development professionals in making evidence-based decisions for clinical-grade MSC manufacturing.
Comparative Performance Analysis: TCP vs. Bioreactor-Expanded MSCs
Table 1: Functional Characteristics of Cryopreserved MSCs Expanded in Different Systems
| Functional Parameter | TCP-Expanded MSCs | Bioreactor-Expanded MSCs | Assessment Method | Post-Thaw Change |
|---|---|---|---|---|
| Adipogenic Differentiation | Maintained [4] | Maintained [4] | Oil Red O staining | No significant difference between systems [4] |
| Osteogenic Differentiation | Maintained [4] [76] | Maintained [4] | Alizarin Red staining | No significant difference between systems [4] |
| Chondrogenic Differentiation | Maintained [4] [76] | Maintained [4] | Alcian Blue/Toluidine Blue staining | No significant difference between systems [4] |
| Colony-Forming Efficiency (CFU-F) | Maintained, though may be lower than HFB [4] | Maintained, potentially higher than TCP [4] | Crystal violet staining of colonies | Trend toward higher potential in HFB, not statistically significant [4] |
| Surface Marker Expression (CD105) | Significant decrease post-thaw (≈75% positive) [4] | Maintained post-thaw (>95% positive) [4] | Flow Cytometry | Significant difference between systems post-thaw [4] |
| Surface Marker Expression (CD44, CD90, CD73) | CD44 may decrease immediately post-thaw [76] | Generally maintained [6] | Flow Cytometry | Acclimation period (24h) restores CD44 in TCP [76] |
| Proliferation / Population Doubling Time | Variable post-thaw recovery [76] | Comparable to TCP [4] | Metabolic activity assays (e.g., Resazurin) | Decreased metabolic activity immediately post-thaw in TCP, recovers after 24h [76] |
| Cell Viability Post-Thaw | >90% [4] | >90% [4] | Trypan Blue/Flow Cytometry | TCP cells may demonstrate greater robustness [4] |
| Immunomodulatory Function | Maintained but may be reduced immediately post-thaw [76] | Maintained (inferred from phenotype) [6] | T-cell proliferation assay | Significantly more potent after 24h acclimation [76] |
Detailed Experimental Protocols for Functional Potency Assessment
Trilineage Differentiation Assay
The trilineage differentiation potential confirms the multipotent character of MSCs after expansion and cryopreservation. The following protocol is standardized for cells post-thaw, with adaptations for specific lineages.
General Protocol Notes:
- Cell Seeding: Use cryopreserved MSCs that have been thawed and allowed a brief acclimation period (recommended 24 hours) to recover [76]. Seed cells at appropriate densities in standard culture plates.
- Induction Media: Replace standard growth medium with specific induction media. Use complete medium without inducers as a negative control.
- Media Refreshment: Change differentiation media every 2-3 days to maintain nutrient and inducer concentrations.
- Fixation and Staining: After the induction period, wash cells with phosphate-buffered saline (PBS), fix with 4% paraformaldehyde, and perform lineage-specific staining.
Lineage-Specific Methodologies:
Adipogenic Differentiation
Osteogenic Differentiation
Chondrogenic Differentiation
- Cell Pellet Formation: Pellet 2.0 × 10⁵ cells in a 15-mL conical tube via gentle centrifugation [77].
- Induction Period: 21 days [77] [4].
- Staining Method: Section pellets and stain with Toluidine Blue [77] or Alcian Blue [4] [76] to detect sulfated proteoglycans in the extracellular matrix.
- Analysis: Qualitative assessment of blue-stained matrix sections under a microscope.
Colony-Forming Unit Fibroblast (CFU-F) Assay
This assay quantifies the clonogenic capacity of MSCs, a key indicator of stemness, following cryopreservation.
- Cell Seeding: Thaw MSCs and seed at low densities (e.g., 100-1000 cells) in a 10-cm culture dish to allow for isolated colony formation [4] [78].
- Culture Conditions: Maintain cells in standard MSC growth medium for 10-14 days without disturbance to allow colony formation from single progenitor cells.
- Staining and Quantification: After the culture period, wash cells with PBS, fix with 4% paraformaldehyde, and stain with 0.5% crystal violet solution for 30 minutes [4]. Wash gently to remove excess stain.
- Analysis: Count the number of colonies manually or with imaging software. A colony is typically defined as a cluster of 50 or more cells [4]. The result is expressed as the colony-forming efficiency (CFE): (Number of colonies formed / Number of cells seeded) × 100%.
Signaling Pathways and Experimental Workflows
Experimental Workflow for Comparative Potency Assessment
The following diagram illustrates the logical sequence for comparing TCP and bioreactor-expanded MSCs post-cryopreservation.
Key Signaling Pathways in MSC Differentiation
The process of trilineage differentiation is governed by complex signaling pathways. The following diagram summarizes the core pathways involved in each lineage commitment, which can be indirectly assessed through the successful execution of the differentiation protocols.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for MSC Potency Assays
| Reagent / Material | Function in Assay | Specific Examples & Notes |
|---|---|---|
| Trilineage Differentiation Kits | Provides optimized, standardized media components for inducing osteogenic, chondrogenic, and adipogenic lineages. | StemPro Differentiation Kits (Thermo Fisher) [76]; Biological Industries kits [77]. |
| Lineage-Specific Stains | Histochemical detection of differentiated cell types. | Oil Red O (lipids) [77] [4]; Alizarin Red S (calcium) [4] [76]; Alcian Blue/Toluidine Blue (proteoglycans) [77] [4]. |
| Cryopreservation Medium | Protects cell viability during freeze-thaw cycle. | Typically 90% FBS + 10% DMSO [77] [76]. For clinical translation, serum-free alternatives and human platelet lysates are investigated [78]. |
| Flow Cytometry Antibody Panels | Immunophenotyping to confirm MSC identity post-thaw per ISCT criteria. | Positive: CD73, CD90, CD105 [77] [4] [76]. Negative: CD34, CD45, CD11b, CD19, HLA-DR [77] [76]. Monitor CD105 decrease in TCP post-thaw [4]. |
| Serum-Free / Xeno-Free Media | Defined culture medium for clinically compliant expansion. | MSC NutriStem XF [78]; StemMACS MSC XF [78]. Supports growth while minimizing batch variability and safety concerns of FBS [78]. |
| Cell Culture Vessels | Platform for cell expansion. | TCP: T-flasks, CellSTACK [63]. Bioreactor: Hollow Fiber Bioreactor (HFB) [4]. Choice influences scalability, labor, and potentially post-thaw function [4]. |
| Metabolic Activity Assay | Assess cell viability and proliferation post-thaw. | Resazurin-based assays (e.g., Vybrant) [76]. Measures reduction of reagent to fluorescent resorufin by viable cells. |
The comparative analysis of TCP and bioreactor-expanded MSCs following cryopreservation reveals a complex landscape. Both expansion systems are capable of producing cells that maintain core functional properties, including trilineage differentiation potential and clonogenicity. However, critical differences emerge, particularly in the preservation of specific surface markers like CD105 in TCP-expanded cells post-thaw and a potential trend toward enhanced colony-forming efficiency in HFB-expanded cells. The evidence strongly supports the implementation of a 24-hour post-thaw acclimation period to recover the full functional potency of cryopreserved MSCs, especially for cells expanded in TCP systems. The choice between TCP and bioreactor systems ultimately depends on the specific requirements of the clinical or research application, weighing factors such as scalability, reproducibility, and the critical need to preserve specific stem cell attributes through the rigors of cryopreservation.
Mesenchymal Stem/Stromal Cells (MSCs) have emerged as a cornerstone of regenerative medicine and immunomodulatory therapy. While initially valued for their differentiation potential, research has demonstrated that their therapeutic benefits are primarily mediated through paracrine signaling rather than direct cell replacement [79] [80]. The collection of bioactive factors secreted by MSCs, termed the "secretome", includes soluble proteins and extracellular vesicles (EVs) that orchestrate immunomodulatory effects on both innate and adaptive immune systems [80] [81].
A critical advancement in this field is the understanding that the composition and potency of the MSC secretome are not fixed but can be significantly influenced by external conditions during cell expansion. This guide provides a detailed comparison of how two fundamental expansion systems—Tissue Culture Polystyrene (TCP) flasks and Hollow Fiber Bioreactors (HFB)—affect the characteristics of MSCs and their resulting secretome, particularly after the critical process of cryopreservation, which is essential for creating "off-the-shelf" therapeutic products [4].
Experimental Comparison: TCP vs. Bioreactor Expansion Post-Cryopreservation
Methodology for Comparative Analysis
To ensure a valid comparison of TCP and HFB systems, a study was designed where adipose-derived stem cells (ASCs) were expanded using equivalent population doublings [4].
- HFB Expansion: A single passage in a Hollow Fiber Bioreactor with a 1.7 m² surface area.
- TCP Expansion: Serial passaging (up to P4) in traditional T175 flasks, theoretically yielding a total of 0.47 m² to match the HFB scale.
- Cryopreservation: Cells from both systems were cryopreserved and then thawed for post-preservation analysis.
- Functional Assays: Post-thaw analyses included immunophenotyping, clonogenicity (CFU), trilineage differentiation, proliferation potential, and a surrogate wound healing assay to test paracrine effects on fibroblast migration [4].
Key Findings: Phenotypic and Functional Differences
The following tables summarize the core experimental data, highlighting the impact of expansion system and cryopreservation on MSC properties.
Table 1: Impact of Expansion System and Cryopreservation on Key Surface Markers
| Surface Marker | Biological Meaning | TCP Pre-Freeze | TCP Post-Thaw | HFB Pre-Freeze | HFB Post-Thaw | Significant Finding |
|---|---|---|---|---|---|---|
| CD105 | Vascular hemostasis; MSC marker [79] | >95% | ~75% | >95% | >95% | Significant decrease only in TCP after thaw [4] |
| CD274 (PD-L1) | Immunomodulatory ligand [82] | Higher | ~48% increase | Significantly Lower | Comparable to TCP | Freeze-thaw balanced initial difference [4] |
| CD73 & CD90 | MSC markers; cell interactions [79] | >95% | >95% | >95% | >95% | Highly expressed, unaffected by system or freeze [4] |
| Stro-1 & CD271 | Primitive/progenitor markers [79] [4] | ~10% (Stro-1) | Consistent | ~10% (Stro-1) | Consistent | No significant change from system or freeze [4] |
Table 2: Comparison of Functional Characteristics Post-Thaw
| Functional Characteristic | TCP-Expanded ASCs | HFB-Expanded ASCs | Statistical Significance |
|---|---|---|---|
| Trilineage Differentiation | Positive (Adipo, Osteo, Chondro) | Positive (Adipo, Osteo, Chondro) | No difference [4] |
| Colony-Forming Unit (CFU) Potential | Robust | Trended Higher | Not Significant [4] |
| Post-Thaw Viability | >90% (More robust) | >90% | TCP demonstrated greater robustness [4] |
| Proliferation Capacity | Unaffected | Unaffected | No difference between systems [4] |
| Effect on Fibroblast Migration | Promoted (Paracrine effect) | Promoted (Paracrine effect) | No statistical difference [4] |
The data reveals that while both systems produce MSCs that meet core functional criteria after cryopreservation, significant differences emerge in specific immunophenotypic subpopulations. The freeze-thaw process itself acts as a selective pressure, driving phenotypic changes. Notably, TCP-expanded cells became less variable, whereas HFB-expanded cells displayed increased heterogeneity after thawing [4]. This highlights that the expansion system is a critical variable determining cellular composition.
Immunomodulatory Mechanisms of the MSC Secretome
The therapeutic secretome, influenced by the expansion method, mediates its effects through a complex network of paracrine signals. The following diagram illustrates the key signaling pathways through which the licensed MSC secretome interacts with and suppresses effector T-cells, while promoting a regulatory immune environment.
Diagram 1: MSC Secretome Immunomodulatory Signaling. The licensed MSC2 phenotype releases a secretome containing soluble factors and extracellular vesicles that collectively suppress effector T-cells, promote anti-inflammatory macrophage polarization, and enhance regulatory T-cell function, leading to controlled inflammation and tissue repair.
Key Soluble Factors and Their Targets
The secretome's immunomodulatory power is executed through a cocktail of key soluble factors:
- Prostaglandin-E2 (PGE2): Suppresses T cell proliferation, inhibits Natural Killer (NK) cell function, promotes the differentiation of regulatory T cells (Tregs), and drives macrophage polarization towards the anti-inflammatory M2 phenotype [81].
- Galectins (e.g., Galectin-9): Galectin-9 interaction with Tim-3 on T cells can induce T-cell suppression and apoptosis of T-helper cells, thereby curbing inflammation [82] [81].
- Indoleamine 2,3-dioxygenase (IDO): This enzyme is upregulated in MSCs by inflammatory cytokines like IFN-γ. IDO catabolizes tryptophan, an essential amino acid for T-cells. Tryptophan depletion and the accumulation of metabolites like kynurenines lead to cell cycle arrest and apoptosis of effector T-cells, while promoting Treg generation [82].
- Growth Factors (HGF, VEGF): Hepatocyte Growth Factor (HGF) and Vascular Endothelial Growth Factor (VEGF) contribute to tissue repair and also possess direct immunomodulatory functions, such as inhibiting T-cell proliferation [82] [81].
Enhancing Secretome Potency: Preconditioning and Licensing
The immunomodulatory profile of the MSC secretome is not static. Its composition can be "licensed" or optimized through preconditioning, a process that polarizes MSCs towards a potent anti-inflammatory (MSC2) phenotype [82] [81]. The following workflow outlines a systematic protocol for licensing MSCs to maximize the immunomodulatory quality of the derived secretome.
Diagram 2: Experimental Workflow for Licensing MSCs and Producing an Immunomodulatory Secretome. A standardized protocol for licensing MSCs using a combination of IFN-γ and TNF-α to generate a secretome with enhanced immunomodulatory properties.
A 2025 study systematically optimized this licensing strategy and found that overnight treatment with a 1:1 ratio of IFN-γ and TNF-α at 60 ng/mL, followed by a 48-hour secretome production phase at 90% cellular confluence, yielded a conditioned media with significantly enhanced capacity to suppress the activation of human peripheral blood mononuclear cells (PBMCs) [82]. This protocol more than doubled the effectiveness of suboptimal licensing methods.
The Scientist's Toolkit: Key Reagents and Systems
Table 3: Essential Research Tools for MSC Secretome and Immunomodulation Studies
| Tool / Reagent | Function / Application | Example / Note |
|---|---|---|
| Hollow Fiber Bioreactor (HFB) | Automated, scalable expansion of adherent MSCs; closed system for GMP compliance [4] [5]. | Quantum Cell Expansion System (Terumo BCT) [5]. |
| Xeno-Free/Sera-Free Media | Defined, clinical-grade medium for MSC expansion without animal serum [32]. | PRIME-XV MSC Expansion XSFM [32]. |
| DMSO-Free Cryopreservation | Freeze-thaw solution avoiding dimethyl sulfoxide toxicity concerns [32]. | PRIME-XV FreezIS DMSO-Free [32]. |
| Licensing Cytokines | Preconditioning MSCs to enhance secretome's immunomodulatory factor content [82]. | Recombinant Human IFN-γ & TNF-α. |
| Multiplex Cytokine Assay | High-throughput profiling of secretome composition [83] [82]. | Bio-Plex Pro Human Cytokine Assay [83]. |
| Functional Immune Assays | Validate secretome bioactivity on target immune cells. | PBMC suppression assay; Treg induction assay; Macrophage polarization [84] [82]. |
The choice between TCP and HFB expansion systems presents a strategic trade-off for researchers. TCP remains a cost-effective and versatile choice for smaller-scale research, while HFB offers superior scalability, automation, and consistency for clinical-grade manufacturing [4]. Critically, both systems can produce functionally competent MSCs after cryopreservation, albeit with distinct immunophenotypic profiles that may influence their specific therapeutic applications.
The future of secretome-based therapies lies in moving beyond uncharacterized conditioned media. The field is advancing towards the use of defined extracellular vesicle (EV) products and the implementation of precise licensing protocols [80] [82]. This, combined with robust, scalable manufacturing in systems like HFB, will pave the way for the development of potent, consistent, and "off-the-shelf" cell-free therapies for treating immune-mediated inflammatory diseases.
The translation of Mesenchymal Stromal Cell (MSC)-based therapies from laboratory research to clinical applications faces substantial manufacturing challenges, particularly concerning the expansion and preservation of cells without compromising their therapeutic efficacy. For acute conditions where rapid administration is crucial, the availability of cryopreserved "off-the-shelf" MSC products becomes essential [85]. The expansion method—whether in traditional tissue culture polystyrene (TCP) flasks or various bioreactor systems—creates a critical decision point in therapeutic development, potentially influencing cellular characteristics and subsequent in vivo performance [4] [86]. This guide objectively compares the functional efficacy of TCP versus bioreactor-expanded MSCs following cryopreservation, synthesizing current preclinical evidence to inform research and development strategies for researchers, scientists, and drug development professionals.
Comparative Analysis of Post-Thaw MSC Characteristics by Expansion Method
Immunophenotypic Profiles and Cellular Heterogeneity
The immunophenotypic profile of MSCs, a set of quality attributes defined by the International Society for Cellular Therapy, can be influenced by both expansion method and the cryopreservation process.
Table 1: Surface Marker Expression Post-Cryopreservation by Expansion System
| Surface Marker | TCP-Expanded ASCs | HFB-Expanded ASCs | Functional Significance |
|---|---|---|---|
| CD73, CD90 | >95% positive (stable) | >95% positive (stable) | Standard MSC positivity markers [4] |
| CD105 | Significant decrease post-thaw (to ~75%) | >95% positive (stable) | MSC marker; loss may indicate phenotype alteration [4] |
| CD274 (PD-L1) | Significant increase post-thaw (~48%) | Increases to comparable level post-thaw | Immunomodulatory protein [4] |
| CD29, CD201 | ~100% positive (stable) | ~100% positive (stable) | Adhesion and progenitor markers [4] |
| CD36, CD31 | <9% positive (stable) | <9% positive (stable) | Negative MSC markers [4] |
| Stro-1 | ~10% positive (stable) | ~10% positive (stable) | Progenitor marker [4] |
Comparative analysis reveals that while most markers remain consistent across systems post-thaw, critical differences emerge in specific immunomodulatory and identity markers. CD105 expression significantly decreases in TCP-expanded cells after freeze-thawing, whereas HFB-expanded cells maintain stable expression [4]. CD274 (PD-L1), an important immunomodulatory protein, shows significantly lower expression on HFB-expanded cells before freezing, but this difference equilizes post-thaw due to a substantial increase in TCP-expanded cells [4].
Beyond individual markers, the two expansion methods support different immunophenotypic subpopulations, influencing the heterogeneity of the final product. TCP and HFB systems demonstrated distinct patterns in the prevalence of subpopulations defined by co-expression patterns of markers like CD166, CD34, CD146, CD271, CD274, and CD248. After thawing, TCP-expanded cells became less variable in their subpopulation distribution, while HFB-expanded cells became more heterogeneous [4]. This suggests that the freeze-thaw process interacts uniquely with each expansion system, driving differential changes in cellular subpopulations that may impact therapeutic consistency.
Functional Potency and Efficacy Metrics
Functional characteristics determining MSC therapeutic potential—including differentiation capacity, proliferation, clonogenicity, and paracrine effects—show nuanced responses to expansion methods post-cryopreservation.
Table 2: Functional Characteristics of Cryopreserved MSCs by Expansion Method
| Functional Attribute | TCP-Expanded ASCs | HFB-Expanded ASCs | Testing Method |
|---|---|---|---|
| Trilineage Differentiation | Maintained (post-thaw) | Maintained (post-thaw) | Oil Red O (adipogenesis), Alizarin Red S (osteogenesis), Alcian Blue (chondrogenesis) [4] |
| Viability | >90% (robust) | >90% | Trypan blue exclusion [4] |
| Population Doubling Time | No significant difference | No significant difference | Growth kinetics over culture period [4] |
| Colony-Forming Unit (CFU) Potential | Present (lower trend, NS) | Present (higher trend, NS) | Colony-forming unit assay [4] |
| Wound Healing (Paracrine) Effects | Supported fibroblast migration | Supported fibroblast migration | Scratch assay using human dermal fibroblasts [4] |
Despite the immunophenotypic differences noted in Table 1, the core functional characteristics of MSCs remain largely intact after cryopreservation regardless of expansion system. Both TCP and HFB-expanded cells retain trilineage differentiation capacity, demonstrating their ability to differentiate into adipocytes, osteoblasts, and chondrocytes after thawing [4]. Similarly, proliferation kinetics and viability post-thaw show no significant differences between systems, with both maintaining viability exceeding 90% [4].
Therapeutically relevant paracrine functions also appear preserved. In wound healing assays designed to assess the granulation phase of repair, both TCP and HFB-expanded MSCs demonstrated comparable ability to support human dermal fibroblast migration, suggesting maintained paracrine functionality critical for many MSC therapeutic applications [4]. This functional preservation across expansion platforms is supported by a systematic review of preclinical inflammation models, which found that the majority (97.7%) of in vivo efficacy outcomes showed no significant difference between freshly cultured and cryopreserved MSCs [87] [85].
Experimental Methodologies for Comparative Assessment
Standardized Expansion and Cryopreservation Protocols
To ensure valid comparisons between expansion systems, researchers must implement standardized protocols that account for fundamental differences in culture parameters and handling.
Hollow Fiber Bioreactor (HFB) Expansion: In comparative studies, one-fifth of ASCs are typically seeded in an HFB system with approximately 1.7 m² surface area for a single passage. Cells are cultured in MSC-qualified media, often supplemented with fetal bovine serum (FBS) or human platelet lysate (HPL), with continuous monitoring of glucose consumption and waste product accumulation [4] [88].
TCP Flask Expansion: For equivalent comparison, fourth-fifth of ASCs (equivalent to a quarter of HFB-cell numbers) are seeded into T175 TCP flasks (0.175 m² each) and expanded through multiple passages (typically to P4) with 1:3 splitting, theoretically yielding 27 T175 flasks (totaling 4.47 m²) [4]. In practice, for manageable scale, one-third of TCP cells may be continued in each subsequent passage.
Controlled-Rate Cryopreservation: Both HFB (at P1) and TCP (at P4) cells are cryopreserved using standardized protocols. Cells are resuspended in cryoprotectant medium, typically containing 10% DMSO and animal-derived product, then frozen using controlled-rate freezers at approximately -1°C/minute to -80°C before transfer to liquid nitrogen storage [4] [6]. For thawing, vials are rapidly warmed in a 37°C water bath and immediately diluted in pre-warmed culture medium to minimize DMSO toxicity.
Preclinical Efficacy Testing in Inflammation Models
Preclinical assessment of MSC therapeutic efficacy typically employs standardized inflammation models that replicate human disease pathophysiology.
Animal Model Selection: Studies commonly use immunocompetent rodent models of inflammation including acute lung injury, sepsis, myocardial infarction, graft-versus-host disease, and inflammatory bowel disease. Immunocompromised models (e.g., SCID mice) are typically excluded as an intact immune system may be required for MSC immunomodulation via host cytotoxic cell activity [85].
MSC Administration Protocols: Cryopreserved MSCs are typically thawed and administered immediately or after less than 24 hours of post-thaw culture, while freshly cultured MSCs serve as comparisons. Cells are delivered via routes relevant to the disease model (intravenous, intraperitoneal, or local injection) at clinically relevant doses (typically 2-10×10⁶ cells/kg) [85] [63].
Efficacy Endpoint Measurement: Primary outcomes include disease-specific metrics such as mortality rates, histological injury scores, inflammatory cytokine levels (TNF-α, IL-6, IL-1β), functional recovery parameters, and biochemical markers of organ dysfunction. Secondary in vitro potency measures may include immunomodulatory capacity (T-cell suppression assays), secretory profiles, and mitochondrial transfer potential [85].
Visualizing Experimental Workflows and Cellular Dynamics
Comparative Analysis Workflow diagrams the standardized experimental approach for comparing TCP and bioreactor-expanded MSCs after cryopreservation, highlighting parallel processing paths and assessment endpoints.
Phenotypic and Functional Relationships illustrates the connection between specific immunophenotypic alterations and functional outcomes, highlighting that despite marker expression variations, core therapeutic functions remain intact across expansion platforms.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for Comparative MSC Studies
| Reagent/Material | Specification/Function | Application Notes |
|---|---|---|
| Hollow Fiber Bioreactor | Spectrum Laboratories; 28 cm² surface area, 0.5-1.5 μm pore size [4] [88] | Enables scalable 3D expansion; maintains cell-matrix interactions |
| Tissue Culture Polystyrene Flasks | T175 flasks (0.175 m² each); Falcon or equivalent [4] [86] | Standard 2D expansion; hydrophilic surface enhances cell adhesion |
| Cryoprotectant Medium | 10% DMSO in FBS or serum-free alternatives [4] [6] | Prevents intracellular ice formation during freezing |
| Flow Cytometry Antibodies | CD73, CD90, CD105, CD274, CD34, CD45 [4] | ISCT phenotype confirmation plus additional characterization |
| Trilineage Differentiation Kits | Adipogenic (Oil Red O), Osteogenic (Alizarin Red S), Chondrogenic (Alcian Blue) [4] [86] | Functional potency verification post-thaw |
| Controlled-Rate Freezer | -1°C/minute cooling rate to -80°C [4] [6] | Standardized freezing protocol for reproducibility |
| Animal Inflammation Models | Immunocompetent rodents (ALI, sepsis, GvHD, IBD) [85] | In vivo efficacy assessment in context of intact immunity |
The collective evidence from preclinical studies indicates that both TCP and bioreactor expansion systems can produce cryopreserved MSC products that retain core therapeutic functions, despite nuanced differences in immunophenotypic profiles and subpopulation distributions. The maintenance of trilineage differentiation capacity, proliferation potential, and paracrine functions across both platforms suggests that expansion method selection may be guided by practical manufacturing considerations rather than definitive efficacy advantages [4] [87].
For clinical translation, this evidence base provides crucial flexibility in manufacturing strategy. Bioreactor systems offer advantages in scalability and consistency for large-scale production, while TCP systems remain accessible for smaller-scale or research applications [4] [63]. The demonstration that cryopreservation does not fundamentally compromise MSC functionality supports the feasibility of "off-the-shelf" therapeutic products for acute inflammatory conditions where rapid administration is essential [85]. Future research should focus on optimizing cryopreservation protocols specific to expansion systems and further elucidating the clinical significance of the observed immunophenotypic differences in specific disease contexts.
Conclusion
The choice between TCP and bioreactor expansion systems has profound implications for the phenotype and function of MSCs after cryopreservation. While TCP remains accessible, bioreactors like the hollow fiber system offer superior scalability, reduced manual handling, and can generate cells with comparable or enhanced functional potency post-thaw, despite driving different immunophenotypic subpopulations. Key challenges remain in optimizing cryopreservation protocols to minimize CPA toxicity and maintain consistent therapeutic efficacy. Future directions must focus on integrating advanced manufacturing with tailored cryopreservation strategies, adopting serum-free media, and employing robust potency assays to ensure the production of reliable, clinically effective 'off-the-shelf' MSC therapies. Collaborative efforts between academia, industry, and regulators are essential to standardize these processes and fully realize the potential of MSC-based regenerative medicine.
References
- 1. The Immunomodulatory mechanism and research progress of ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04333-2]
- 2. Efficient Manufacturing of Therapeutic Mesenchymal Stromal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4087082/]
- 3. Mesenchymal Stem Cell Immunomodulation: Mechanisms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7751844/]
- 4. Comparative analysis of cryopreserved adipose stem cells ... [https://www.nature.com/articles/s41598-024-83255-0]
- 5. A comprehensive review of quantum bioreactor cell ... [https://www.sciencedirect.com/science/article/pii/S146532492300097X]
- 6. Enhanced cryopreservation of MSCs in microfluidic ... [https://www.nature.com/articles/srep35416]
- 7. Current perspectives on the dynamic culture of mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11954588/]
- 8. Mesenchymal Stem Cells Market Report 2025–2035 [https://www.marketbusinessinsights.com/mesenchymal-stem-cells-market]
- 9. mesenchymal stem cells market size and share analysis [https://www.coherentmarketinsights.com/market-insight/mesenchymal-stem-cells-market-720]
- 10. Mesenchymal stem cells in treating human diseases [https://www.nature.com/articles/s41392-025-02313-9]
- 11. Mesenchymal stem cells: opening a new chapter in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12465317/]
- 12. Human Adipose-Derived Mesenchymal Stem Cells ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4770156/]
- 13. Short-Term Cryopreservation Preserved the Function of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12524150/]
- 14. Comparison of 3D culture systems on mesenchymal stem cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12562725/]
- 15. Osteogenic differentiation of human mesenchymal stem ... [https://www.sciencedirect.com/science/article/pii/S2405844024049119]
- 16. Mesenchymal Stem Cells/Medicinal Signaling ... [https://www.businesswire.com/news/home/20251009529610/en/Mesenchymal-Stem-CellsMedicinal-Signaling-Cells-Advances-Applications-Report-2025-1670-Trials-12-Approvals-Worldwide-and-Path-to-50-Approvals-by-2040---ResearchAndMarkets.com]
- 17. From bench to bedside: translating mesenchymal stem cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12344367/]
- 18. Cell retention in scalable, perfusion-based mesenchymal ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1611703/full]
- 19. Key quality parameter comparison of mesenchymal stem ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2024.1412811/full]
- 20. Identification of critical process parameters and quality ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12408662/]
- 21. Effects, methods and limits of the cryopreservation on ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-024-03954-3]
- 22. Cryopreservation as a Key Element in the Successful Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7727450/]
- 23. Taking lessons from nature to improve cell therapy ... [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/2759/taking-lessons-from-nature-to-improve-cell-therapy-cryopreservation]
- 24. Magnetic heating of nanoparticles as a scalable ... [https://www.nature.com/articles/s41598-020-70707-6]
- 25. A comparison of bioreactors for culture of fetal ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961210009695]
- 26. A New Perspective for Bone Tissue Engineering [https://www.mdpi.com/1422-0067/23/3/1425]
- 27. Reassessing Long-Term Cryopreservation Strategies for ... [https://www.sciencedirect.com/science/article/pii/S0022202X24018980]
- 28. The Future of Networking: Emerging Protocols to Watch in 2025 [https://blog.j2sw.com/networking/the-future-of-networking-emerging-protocols-to-watch-in-2025/]
- 29. draft-ietf-tcpm-tcp-edo-15 - TCP Extended Data Offset Option [https://datatracker.ietf.org/doc/draft-ietf-tcpm-tcp-edo/]
- 30. A dozen extensions to TCP/IP that optimize internet ... [https://www.networkworld.com/article/956346/a-dozen-extensions-to-tcp-ip-that-optimize-internet-connections.html]
- 31. TCP SYN Extended Option Space Using an Out-of-Band ... [https://datatracker.ietf.org/doc/draft-touch-tcpm-tcp-syn-ext-opt/]
- 32. Advancing Key Processes of the MSC Therapy ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324925001902]
- 33. Comparative Analysis of Hollow-Fiber and Traditional ... [https://hollow-fiber.com/blog/comparative-analysis-of-hollow-fiber-and-traditional-bioreactors-in-cell-culture/]
- 34. Comparison of BHK-21 cell growth on microcarriers vs in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3980977/]
- 35. Comparisons of microcarrier cell culture processes in one ... [https://link.springer.com/article/10.1007/s004490050544]
- 36. Hollow fiber membrane bioreactors for efficient CO2 ... [https://www.sciencedirect.com/science/article/pii/S0960852425006819]
- 37. Enhanced cryopreservation of MSCs in microfluidic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5066325/]
- 38. Top 6 Single Use Bioreactor Companies to Watch in 2025 [https://www.expertmarketresearch.com/healthcare-articles/top-companies-in-the-single-use-bioreactor-market]
- 39. Automated Manufacturing Processes and Platforms for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11872983/]
- 40. Automated manufacturing of cell therapies [https://www.sciencedirect.com/science/article/pii/S0168365925001701]
- 41. Why Cell Therapy Systems Are Going Automatic: A New ... [https://biopharmaapac.com/report/134/6254/why-cell-therapy-systems-are-going-automatic-a-new-era-of-manufacturing.html]
- 42. Cocoon® Platform | Cell Therapy Manufacturing [https://www.lonza.com/specialized-modalities/cell-and-gene/cocoon-platform]
- 43. Automated production of gene-modified chimeric antigen ... [https://www.sciencedirect.com/science/article/pii/S1465324923010137]
- 44. When using a closed and automated manufacturing ... [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/2156/When-using-a-closed-and-automated-manufacturing-platform-is-there-an-option-to-maintain-flexibility]
- 45. Comparison of culture media supplements identifies serum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12382287/]
- 46. Promoting ethical and reproducible cell culture [https://www.frontiersin.org/journals/toxicology/articles/10.3389/ftox.2025.1670513/full]
- 47. Re-imagining human cell culture media: Challenges, ... [https://www.sciencedirect.com/science/article/abs/pii/S0734975025000503]
- 48. A Chemically Defined, Xeno- and Blood-Free Culture ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2021.619930/full]
- 49. High throughput morphological screening identifies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11889916/]
- 50. The influence of xeno-free culture conditions on ... [https://www.sciencedirect.com/science/article/pii/S2352320424001743]
- 51. Cell retention in scalable, perfusion-based mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12271218/]
- 52. Vitrification versus slow freezing for human ovarian tissue ... [https://www.nature.com/articles/s41598-017-09005-7]
- 53. Comparative effectiveness of vitrification and slow freezing ... [https://bmcwomenshealth.biomedcentral.com/articles/10.1186/s12905-024-03505-1]
- 54. Cryopreservation of human embryos by vitrification or slow ... [https://www.sciencedirect.com/science/article/pii/S0015028207012617]
- 55. Viable and Functional: Long-Term −80 °C Cryopreservation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12525932/]
- 56. A comparative analysis of vitrification and two slow freezing ... [https://link.springer.com/article/10.1007/s44164-025-00085-8]
- 57. Comparison between Slow Freezing and Vitrification in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10374242/]
- 58. Dimethyl sulfoxide: a central player since the dawn of ... [https://pubmed.ncbi.nlm.nih.gov/32342730/]
- 59. Cryoprotectant Toxicity: Balancing Safety and Efficacy [https://allanchem.com/cryoprotectant-toxicity-safety-efficacy/]
- 60. DMSO induces drastic changes in human cellular ... [https://www.nature.com/articles/s41598-019-40660-0]
- 61. Bio-based cryoprotectants, mechanisms, and advanced ... [https://www.sciencedirect.com/science/article/pii/S0924224425003000]
- 62. Cell Cryoprotectant Strategic Roadmap: Analysis and ... [https://www.archivemarketresearch.com/reports/cell-cryoprotectant-328193]
- 63. Large-Scale Expansion of Human Mesenchymal Stem Cells [https://pmc.ncbi.nlm.nih.gov/articles/PMC7378617/]
- 64. Underlying Mechanisms and Treatment of Cellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10917536/]
- 65. Future Perspectives in Senescence-Based Therapies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12190458/]
- 66. Immunosenescence: molecular mechanisms and diseases [https://www.nature.com/articles/s41392-023-01451-2]
- 67. Strategies to enhance stability of cryopreservation ... [https://www.sciencedirect.com/science/article/abs/pii/S0734975025002496]
- 68. Comparative Analysis of Serum and Serum-Free Medium ... [https://www.mdpi.com/1422-0067/25/19/10627]
- 69. Hydrogel microcapsulation technology reduces the DMSO ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12639263/]
- 70. Rapid and Continuous Cryopreservation of Stem Cells with ... [https://www.mdpi.com/2072-666X/13/9/1516]
- 71. Clinical experience with cryopreserved mesenchymal stem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11947892/]
- 72. Chromosomal stability of mesenchymal stromal cells ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324915011470]
- 73. Enhancement of the function of mesenchymal stem cells by ... [https://www.sciencedirect.com/science/article/pii/S2405844024069998]
- 74. Reconstitution and post-thaw storage of cryopreserved ... [https://www.sciencedirect.com/science/article/pii/S2352320423000275]
- 75. CD73, CD90, CD105 and Cadherin-11 RT-PCR Screening ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5984056/]
- 76. Cryopreserved mesenchymal stem cells regain functional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6716839/]
- 77. Effects of cryopreservation and long-term culture on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7247169/]
- 78. Expansion and characterization of bone marrow derived ... [https://www.nature.com/articles/s41598-021-83088-1]
- 79. Mesenchymal Stem/Stromal Cells and Their Paracrine Activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8875256/]
- 80. Mesenchymal stem cell secretome for regenerative medicine [https://www.sciencedirect.com/science/article/pii/S2090123224001814]
- 81. Mesenchymal Stromal Cell Secretome for the Treatment of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8617629/]
- 82. Fine-tuning licensing strategies to boost MSC-based ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04315-4]
- 83. Chemoresistant tumor cell secretome potentiates immune ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12257726/]
- 84. Secretome screening reveals immunomodulating functions ... [https://www.nature.com/articles/s41598-021-96184-z]
- 85. Comparison of freshly cultured versus cryopreserved ... [https://elifesciences.org/articles/75053]
- 86. Concise Review: Optimizing Expansion of Bone Marrow ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4006491/]
- 87. Comparison of freshly cultured versus cryopreserved ... [https://pubmed.ncbi.nlm.nih.gov/35838024/]
- 88. Mesenchymal stromal cell delivery via an ex vivo ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8133341/]